
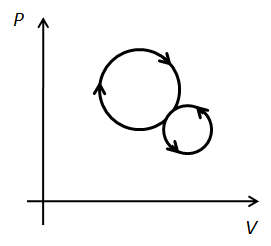
A thermodynamic process undergoes a cyclic process as shown in figure. Over one complete cycle, work done by the system is…… .
A. 0
B.> 0
C. < 0
D. Nothing can be said

Answer
579.9k+ views
Hint: The work is done by the system for the clockwise cycle and the work is done on the system for the anticlockwise cycle. The area under the curve in the PV diagram is the work done.
Complete step by step answer:According to first law thermodynamics, the total internal energy change is the sum of heat energy and work done.
\[\Delta E = Q + W\]
In a cyclic process, the initial and final internal energy of the system is the same. Therefore, the change in internal energy is zero.
\[0 = Q + W\]
\[ \Rightarrow W = - Q\]
Therefore, in a cyclic process, the work done is equal to heat absorbed by the system.
The total work done by the system in the cyclic process is the area under the curve of the PV diagram. For a clockwise cycle, the work is done by the system on the surrounding and for an anticlockwise cycle, surrounding does work on the system.
In the given diagram, the clockwise cycle represents work done by the system and the anticlockwise system represents work done on the system. But the area under the curve for the clockwise cycle is greater than the area under the curve for the anticlockwise cycle. Therefore, the net work will be done by system and it is greater than zero.
So, the correct answer is option (B).
Note:The work done for the cycle process is given as, \[W = \int {PdV} \]. The integration represents the area under the curve. The work done by the system will be negative if the work done on the system is greater than the work done by the system. The work done by the system is zero when the heat absorbed by the system is zero.
Complete step by step answer:According to first law thermodynamics, the total internal energy change is the sum of heat energy and work done.
\[\Delta E = Q + W\]
In a cyclic process, the initial and final internal energy of the system is the same. Therefore, the change in internal energy is zero.
\[0 = Q + W\]
\[ \Rightarrow W = - Q\]
Therefore, in a cyclic process, the work done is equal to heat absorbed by the system.
The total work done by the system in the cyclic process is the area under the curve of the PV diagram. For a clockwise cycle, the work is done by the system on the surrounding and for an anticlockwise cycle, surrounding does work on the system.
In the given diagram, the clockwise cycle represents work done by the system and the anticlockwise system represents work done on the system. But the area under the curve for the clockwise cycle is greater than the area under the curve for the anticlockwise cycle. Therefore, the net work will be done by system and it is greater than zero.
So, the correct answer is option (B).
Note:The work done for the cycle process is given as, \[W = \int {PdV} \]. The integration represents the area under the curve. The work done by the system will be negative if the work done on the system is greater than the work done by the system. The work done by the system is zero when the heat absorbed by the system is zero.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




