
Amongst the following the most basic compound is:
A. p-nitroaniline
B. Acetanilide
C. Aniline
D. Benzylamine
Answer
525.1k+ views
Hint: Amines are bases due to the presence of a lone pair of electrons on the nitrogen atom. The tendency of nitrogen atoms to share these electrons with acid is responsible for the basic strength of amines.
Complete step-by-step answer:
The structure of the compounds are given below;
As we know that the N containing compounds depends on the availability of lone pairs of electrons on N.
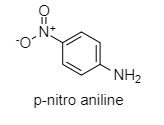
p-nitroaniline: Nitro group at p-position pulls the lone pair of electrons of N more strongly than aniline. Hence, lone pairs of electrons are not available.
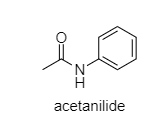
Acetanilide: In this compound, there are two side pulling of lone pairs of electrons of N. One side by benzene ring and the other side by ${ C=O }$ group. So, the lone pair of electrons on N are not available.
Aniline: Lone pairs of electrons on N are involved in resonance with an aromatic benzene ring. Hence, lone pairs are not available easily.
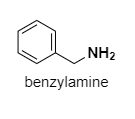
Benzylamine: Here, the lone pairs are not in conjugation with the benzene right. It is very much like a primary amine and lone pair of electrons on N are easily available.
Hence, the most basic compound is Benzylamine and the correct option is D.
Note: The possibility to make a mistake is that you may choose C. But benzylamine is more basic than aniline as the benzyl group is an electron-donating group due to the ${ +I }$ effect. So, the electron density increases on N of ${ NH }_{ 2 }$.
Complete step-by-step answer:
The structure of the compounds are given below;
As we know that the N containing compounds depends on the availability of lone pairs of electrons on N.




p-nitroaniline: Nitro group at p-position pulls the lone pair of electrons of N more strongly than aniline. Hence, lone pairs of electrons are not available.
Acetanilide: In this compound, there are two side pulling of lone pairs of electrons of N. One side by benzene ring and the other side by ${ C=O }$ group. So, the lone pair of electrons on N are not available.
Aniline: Lone pairs of electrons on N are involved in resonance with an aromatic benzene ring. Hence, lone pairs are not available easily.
Benzylamine: Here, the lone pairs are not in conjugation with the benzene right. It is very much like a primary amine and lone pair of electrons on N are easily available.
Hence, the most basic compound is Benzylamine and the correct option is D.
Note: The possibility to make a mistake is that you may choose C. But benzylamine is more basic than aniline as the benzyl group is an electron-donating group due to the ${ +I }$ effect. So, the electron density increases on N of ${ NH }_{ 2 }$.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




