
Benzyl chloride $\left( {{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl \right)$ can be prepared from toluene by chlorination with?
A. $S{{O}_{2}}C{{l}_{2}}$
B. $SOC{{l}_{2}}$
C. $C{{l}_{2}}$
D. NaOCI
Answer
573.3k+ views
Hint: Benzyl chloride$\left( {{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl \right)$ can be prepared from toluene by chlorination with $C{{l}_{2}}$. This is because it is found that in the presence of sunlight, $C{{l}_{2}}$ gives free radicals. This reaction is basically a free radical substitution reaction.
Complete answer:
- We can see that in toluene there is no electrophilic centre, hence oxidation takes place in the presence of sunlight using $C{{l}_{2}}$.
- $S{{O}_{2}}C{{l}_{2}}$ is found to give chlorination, when there is a nucleophilic substitution reaction on an electrophilic centre. But as we know, there is no electrophilic centre in toluene, therefore, chlorination occurs by a controlled free radical mechanism.
- NaOCI Is found to give substitution reaction, and doesn’t give free radical so it will take part in chlorination.
- We can see the chlorination reaction that is, toluene by chlorination with $C{{l}_{2}}$ will give Benzyl chloride as:
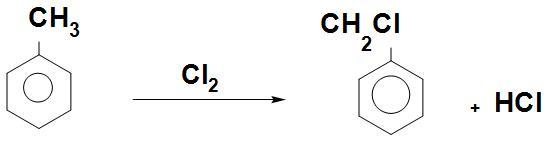
- Benzyl chloride is used in large scale in making resins, dyes, lubricants, drugs and cosmetics.
- Hence, we can conclude that the correct option is (c), that is Benzyl chloride$\left( {{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl \right)$ can be prepared from toluene by chlorination with $C{{l}_{2}}$.
Note: - Industrially, Benzyl chloride can be prepared from toluene and chlorine by the gas phase photochemical mechanism. During this reaction HCl is obtained as a side product.
- It is also found that Benzyl chloride has a strong smell, inflammable to skin and can cause tearing of eyes.
Complete answer:
- We can see that in toluene there is no electrophilic centre, hence oxidation takes place in the presence of sunlight using $C{{l}_{2}}$.
- $S{{O}_{2}}C{{l}_{2}}$ is found to give chlorination, when there is a nucleophilic substitution reaction on an electrophilic centre. But as we know, there is no electrophilic centre in toluene, therefore, chlorination occurs by a controlled free radical mechanism.
- NaOCI Is found to give substitution reaction, and doesn’t give free radical so it will take part in chlorination.
- We can see the chlorination reaction that is, toluene by chlorination with $C{{l}_{2}}$ will give Benzyl chloride as:

- Benzyl chloride is used in large scale in making resins, dyes, lubricants, drugs and cosmetics.
- Hence, we can conclude that the correct option is (c), that is Benzyl chloride$\left( {{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl \right)$ can be prepared from toluene by chlorination with $C{{l}_{2}}$.
Note: - Industrially, Benzyl chloride can be prepared from toluene and chlorine by the gas phase photochemical mechanism. During this reaction HCl is obtained as a side product.
- It is also found that Benzyl chloride has a strong smell, inflammable to skin and can cause tearing of eyes.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




