
Convert chloro benzene to phenol.
Answer
519.6k+ views
Hint: We know that Dow's process is one of the ways of conversion of chlorobenzene to phenol. To obtain phenol from chlorobenzene, first we have to react with chlorobenzene with sodium hydroxide, then phenoxide ion forms. The acidification of phenoxide ions results in phenol.
Complete answer:
In Dow's process, hydrolysis of chlorobenzene results in phenol. First step is treating chlorobenzene with aqueous sodium hydroxide at a temperature of 623 K and pressure of 300 bar to convert it into sodium phenoxide.
Now, we write the reaction of formation of sodium phenoxide ion by reaction of chlorobenzene with sodium hydroxide.

The next step is the acidification of phenoxide ions. This can be done by reacting phenoxide ion with hydrochloric acid. The result of this reaction is the production of phenol.
Now, we write the reaction of production of phenol by acidification of phenoxide ions.

Hence, by Dow’s process, we are able to convert chloro benzene to phenol by Dow’s process.
Additional information:
When one hydrogen atom of the benzene ring is replaced by a hydroxide ion, then the compound is known as phenol. Phenols are commonly used as intermediates in industrial synthesis and in household products. For example, phenol is used as a disinfectant (in low concentrations) in household and mouthwash cleaners. Phenol was probably the first antiseptic surgery. Phenol is also known as carbonic acid, hydroxybenzene and phenolic acid.
Note:
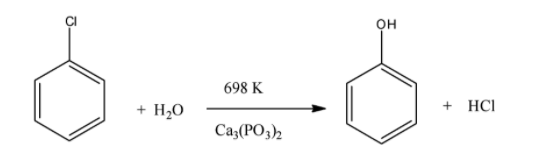
Another way to convert chlorobenzene to phenol is by reacting phenol with steam at 698 K in presence of calcium phosphate. The reaction gives phenol and hydrochloric acid.
Complete answer:
In Dow's process, hydrolysis of chlorobenzene results in phenol. First step is treating chlorobenzene with aqueous sodium hydroxide at a temperature of 623 K and pressure of 300 bar to convert it into sodium phenoxide.
Now, we write the reaction of formation of sodium phenoxide ion by reaction of chlorobenzene with sodium hydroxide.

The next step is the acidification of phenoxide ions. This can be done by reacting phenoxide ion with hydrochloric acid. The result of this reaction is the production of phenol.
Now, we write the reaction of production of phenol by acidification of phenoxide ions.

Hence, by Dow’s process, we are able to convert chloro benzene to phenol by Dow’s process.
Additional information:
When one hydrogen atom of the benzene ring is replaced by a hydroxide ion, then the compound is known as phenol. Phenols are commonly used as intermediates in industrial synthesis and in household products. For example, phenol is used as a disinfectant (in low concentrations) in household and mouthwash cleaners. Phenol was probably the first antiseptic surgery. Phenol is also known as carbonic acid, hydroxybenzene and phenolic acid.
Note:
Another way to convert chlorobenzene to phenol is by reacting phenol with steam at 698 K in presence of calcium phosphate. The reaction gives phenol and hydrochloric acid.

Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions (2025-26)

Solutions Class 12 Chemistry Chapter 1 CBSE Notes - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The d and f Block Elements (2025-26)

Biomolecules Class 12 Chemistry Chapter 10 CBSE Notes - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules (2025-26)




