
Dative bond is present in the molecule:
(A) $N{H_3}$
(B) $C{O_2}$
(C) $NaCl$
(D) None of there
Answer
582.3k+ views
Hint:
Dative bond is a coordinate covalent bond in which two electrons derive from the same atom.
Example: The bonding of metal ions to ligands involves the dative bond.
Complete Step by Step Solution:
Let us draw between the given molecules.
$N{H_3}$molecule: Central atom of $N{H_3}$is nitrogen; it has 5 electron forms covalent bond with their H-atoms. Two electrons remain unbonded. These are available for formation of dative bonds.
Example:
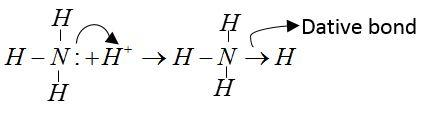
$C{O_2}$ molecule: Central atom is carbon; it has 4 valence electrons. All electrons form a covalent bond with an O-atom.

C-atoms do not have lone pairs of electrons for formation of dative bonds.
$NaCl$ molecule: $NaCl$ is ionic molecule. It forms an ionic bond.
As follows.
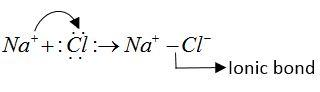
$Na$ and $Cl$ both atoms do not have lone pairs of electrons that form formation of dative bond.
Therefore, form the above explanation the correct option is (A) $N{H_3}.$
Additional Information:
Dative bond is a chemical bond between two atoms in which only one atom provides a shared pair of electrons for bond formation.
Only those atoms form dative bonds, that have lone pairs of electrons.
Dative bond is represented by an arrowhead that points from donor atom to acceptor.
Reaction between Boron Trifluoride $(B{F_3})$and ammonia $(N{H_3})$is due to formation of dative bonds.
$B{F_3}$is electron deficient and ammonia is electron rich so they form dative bonds.
Note:
Sometimes it is difficult to identify dative bonds in compounds. The complex in coordination compounds is formed by dative bonds.
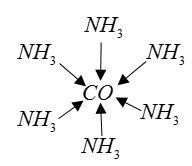
Example: ${[Pt{(N{H_3})_3}Br]^ + }C{l^ - }$complex ion is ${[Pt{(N{H_3})_3}Br]^ + }$dative bond present between metal $Pt$ and ligands $N{H_3}$and $Br.$
Dative bond is a coordinate covalent bond in which two electrons derive from the same atom.
Example: The bonding of metal ions to ligands involves the dative bond.

Complete Step by Step Solution:
Let us draw between the given molecules.
$N{H_3}$molecule: Central atom of $N{H_3}$is nitrogen; it has 5 electron forms covalent bond with their H-atoms. Two electrons remain unbonded. These are available for formation of dative bonds.
Example:
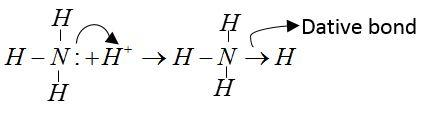
$C{O_2}$ molecule: Central atom is carbon; it has 4 valence electrons. All electrons form a covalent bond with an O-atom.

C-atoms do not have lone pairs of electrons for formation of dative bonds.
$NaCl$ molecule: $NaCl$ is ionic molecule. It forms an ionic bond.
As follows.
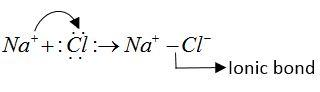
$Na$ and $Cl$ both atoms do not have lone pairs of electrons that form formation of dative bond.
Therefore, form the above explanation the correct option is (A) $N{H_3}.$
Additional Information:
Dative bond is a chemical bond between two atoms in which only one atom provides a shared pair of electrons for bond formation.
Only those atoms form dative bonds, that have lone pairs of electrons.
Dative bond is represented by an arrowhead that points from donor atom to acceptor.
Reaction between Boron Trifluoride $(B{F_3})$and ammonia $(N{H_3})$is due to formation of dative bonds.
$B{F_3}$is electron deficient and ammonia is electron rich so they form dative bonds.
Note:
Sometimes it is difficult to identify dative bonds in compounds. The complex in coordination compounds is formed by dative bonds.
Example: ${[Pt{(N{H_3})_3}Br]^ + }C{l^ - }$complex ion is ${[Pt{(N{H_3})_3}Br]^ + }$dative bond present between metal $Pt$ and ligands $N{H_3}$and $Br.$
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




