
Distinguish between following:
Benzaldehyde and benzoic acid
Answer
573.3k+ views
Hint: Benzaldehyde and benzoic acid both are aromatic compounds, the difference is the group attached to the benzene ring. The functional group for aldehyde is –CHO and the functional group of carboxylic acid is –COOH.
Complete answer:
Benzaldehyde: Benzaldehyde is the simplest aromatic aldehyde which consists of a benzene ring joined with a single formyl group. The other name for benzoic acid is benzenedicarboxaldehyde. By the suffix itself it can be recognized that it is an aldehydic compound.
The structure of benzaldehyde is shown below.
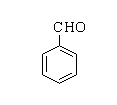
Benzoic acid: Benzoic acid is the simplest aromatic carboxylic acid which consists of a benzene ring joined with a single carboxylic group. The other name for benzoic acid is carboxybenzene. By the suffix –oic itself it can be recognized that it is a carboxylic compound.
The structure of benzoic acid is shown below.
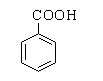
The benzaldehyde can be distinguished from benzoic acid by a sodium bicarbonate test. Benzoic acid is an acid, therefore on reacting with a base sodium bicarbonate it forms a sodium salt of benzoic acid by releasing carbon dioxide gas.
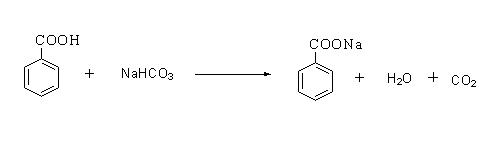
The reaction is shown below.
On the other hand, benzaldehyde does not give any reaction with sodium bicarbonate.
Note:
The other test used to distinguish benzaldehyde and benzoic acid is reacting with Tollen’s reagent also known as silver mirror test. Benzaldehyde reacts with ammoniacal solution of silver nitrate to generate silver mirror.
Complete answer:
Benzaldehyde: Benzaldehyde is the simplest aromatic aldehyde which consists of a benzene ring joined with a single formyl group. The other name for benzoic acid is benzenedicarboxaldehyde. By the suffix itself it can be recognized that it is an aldehydic compound.
The structure of benzaldehyde is shown below.

Benzoic acid: Benzoic acid is the simplest aromatic carboxylic acid which consists of a benzene ring joined with a single carboxylic group. The other name for benzoic acid is carboxybenzene. By the suffix –oic itself it can be recognized that it is a carboxylic compound.
The structure of benzoic acid is shown below.

The benzaldehyde can be distinguished from benzoic acid by a sodium bicarbonate test. Benzoic acid is an acid, therefore on reacting with a base sodium bicarbonate it forms a sodium salt of benzoic acid by releasing carbon dioxide gas.
The reaction is shown below.

On the other hand, benzaldehyde does not give any reaction with sodium bicarbonate.
Note:
The other test used to distinguish benzaldehyde and benzoic acid is reacting with Tollen’s reagent also known as silver mirror test. Benzaldehyde reacts with ammoniacal solution of silver nitrate to generate silver mirror.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Why cannot DNA pass through cell membranes class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE

In a human foetus the limbs and digits develop after class 12 biology CBSE

AABbCc genotype forms how many types of gametes a 4 class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

The correct structure of ethylenediaminetetraacetic class 12 chemistry CBSE




