
Draw electron dot structure of ethane.
Answer
597k+ views
Hint: We can draw electron dot structure easily by calculating the valence electrons on each atom present in ethane. Ethane consists of carbon and hydrogen. In ethane, there is one C-C bond and six C-H bonds.
Complete answer:
Lewis dot structures, also known as electron dot structures, are diagrams that show the bonding between atoms or molecules and the lone pairs of electrons existing in the molecule.
The total number of electrons represented in the electron dot structure is the sum of the total number of valence electrons present in each atom of the molecule.
Electrons other than the valence electrons are not shown in a valence dot structure.
After we have determined the number of valence electrons, we can place them in the dot structure following these steps-
-If we are drawing it for a molecule which consists of two or more atoms, the atoms are connected by single bonds.
-Electrons should be placed as lone pairs. One pair of dots for each pair of electrons should be placed.
-Lone pairs should be placed on outer atoms until each atom has eight electrons. If there are any extra electrons left, they should be placed on the central atom.
-When all the lone pairs are placed, if the central atom did not complete its octet, a double bond should be placed.
We can draw the electron dot structure of ethane as-
Formula of ethane is ${{C}_{2}}{{H}_{6}}$
In ethane, there are two carbon atoms which are singly bonded to each other. On each carbon atom, there are 3 hydrogen atoms.
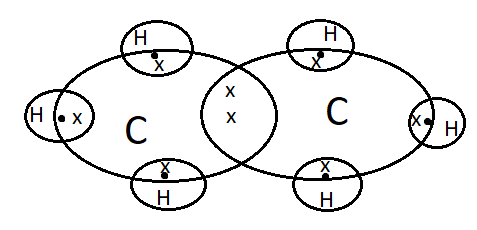 Carbon has 4 valence electrons (represented as ‘x’ in the diagram) and each hydrogen atom has 1 electron (represented a ‘.’ in the diagram)
Carbon has 4 valence electrons (represented as ‘x’ in the diagram) and each hydrogen atom has 1 electron (represented a ‘.’ in the diagram)
1 electron pair is used up in the C-C bond formation.
The remaining 3 electrons are used in bond formation with hydrogen.
Note: To draw electron dot structure, it is important that we remember the structure of compounds like ethane. It is also important to remember the atomic numbers in order to find out the number of valence electrons.
Complete answer:
Lewis dot structures, also known as electron dot structures, are diagrams that show the bonding between atoms or molecules and the lone pairs of electrons existing in the molecule.
The total number of electrons represented in the electron dot structure is the sum of the total number of valence electrons present in each atom of the molecule.
Electrons other than the valence electrons are not shown in a valence dot structure.
After we have determined the number of valence electrons, we can place them in the dot structure following these steps-
-If we are drawing it for a molecule which consists of two or more atoms, the atoms are connected by single bonds.
-Electrons should be placed as lone pairs. One pair of dots for each pair of electrons should be placed.
-Lone pairs should be placed on outer atoms until each atom has eight electrons. If there are any extra electrons left, they should be placed on the central atom.
-When all the lone pairs are placed, if the central atom did not complete its octet, a double bond should be placed.
We can draw the electron dot structure of ethane as-
Formula of ethane is ${{C}_{2}}{{H}_{6}}$
In ethane, there are two carbon atoms which are singly bonded to each other. On each carbon atom, there are 3 hydrogen atoms.

1 electron pair is used up in the C-C bond formation.
The remaining 3 electrons are used in bond formation with hydrogen.
Note: To draw electron dot structure, it is important that we remember the structure of compounds like ethane. It is also important to remember the atomic numbers in order to find out the number of valence electrons.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




