
How can I draw the Lewis dot structure for $Be{{F}_{2}}$ ?
Answer
564.6k+ views
Hint:. Lewis dot structures also known as electron dot structures. Lewis dot structures represent the valence electrons of the atoms present within the molecule. By Lewis dot structures we can visualize the bonding electrons and lone pair of electrons present in the molecule.
Complete step by step answer:
- In the question it is given that to draw the Lewis dot structures of the beryllium difluoride.
- We know that the atomic number of beryllium is 4 and has four electrons in its electronic configuration.
- The electronic configuration of beryllium is $1{{s}^{2}}2{{s}^{2}}$
- So beryllium has two valence electrons in 2s orbital.
- The Lewis dot structure of the beryllium is as follows.
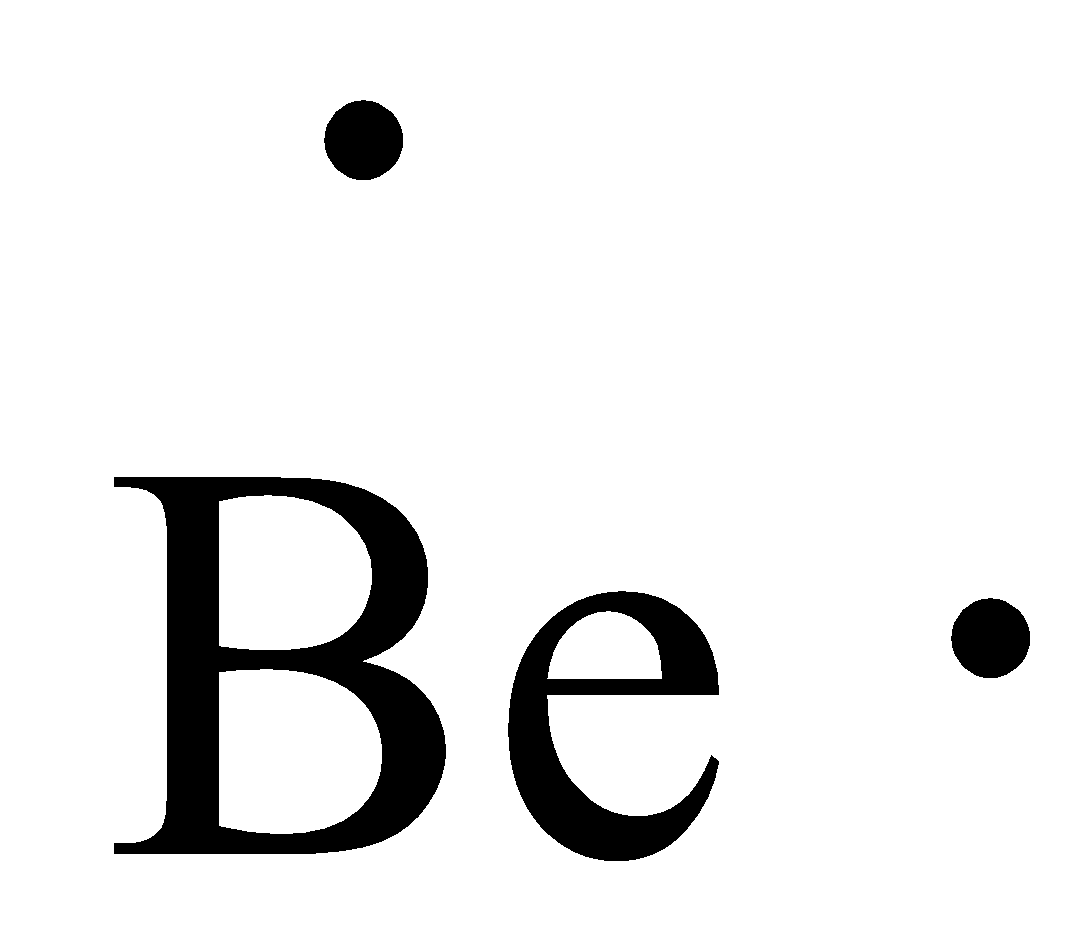
- Coming to fluorine, we know that the fluorine element has seven valence electrons in its electronic configuration.
- The Lewis dot structure of Fluorine is as follows.
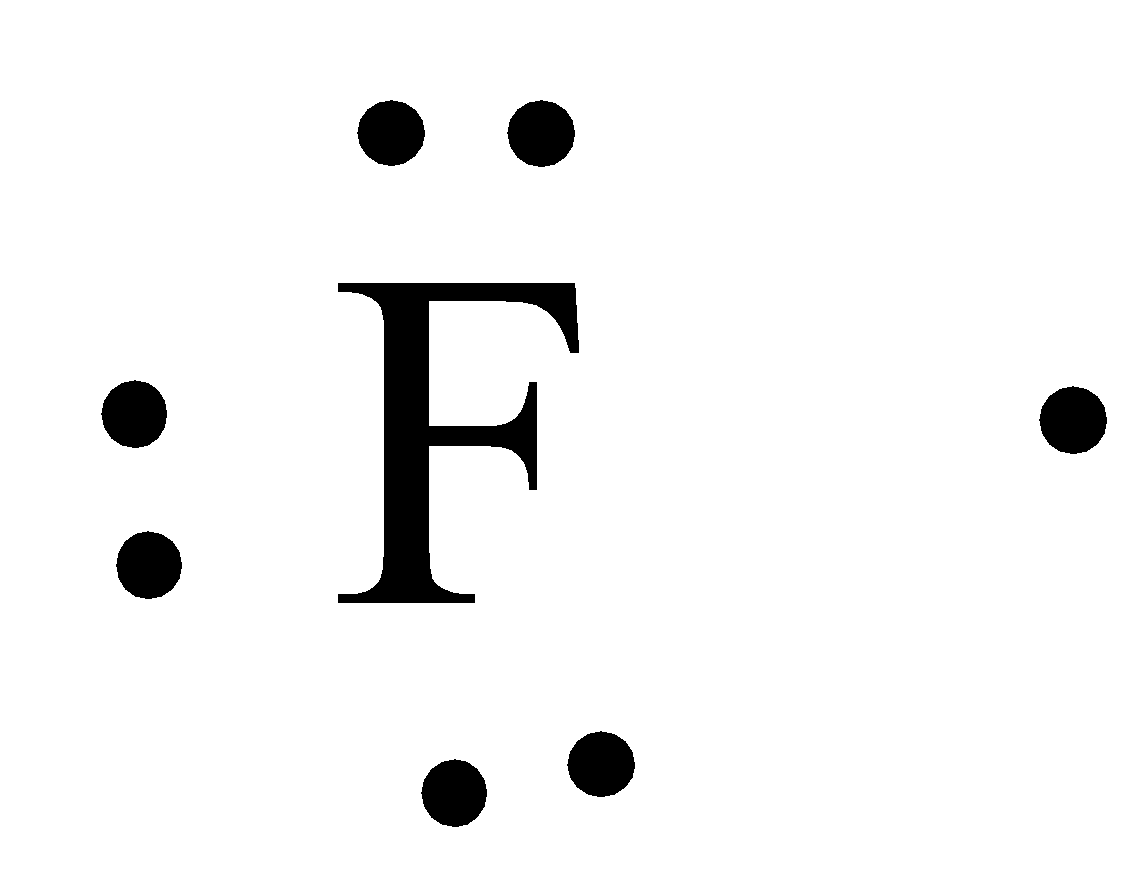
- Now by taking the above structures as examples we can draw the Lewis dot structure of $Be{{F}_{2}}$ and it is as follows.
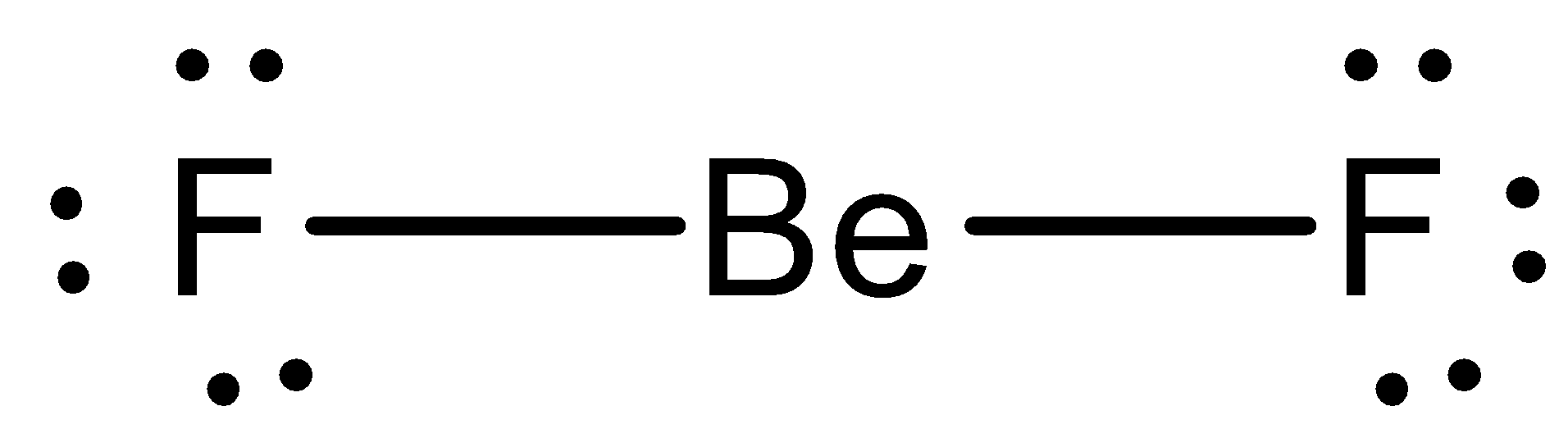
- From the above Lewis dot structure we can easily say that each fluorine atom has three lone pairs of electrons and beryllium has zero lone pair of electrons in Beryllium difluoride molecules.
Note: By using Lewis dot structures of the molecules we can determine the formation of the chemical bonds in various molecules. We can draw the Lewis dot structures for the compounds containing covalent bonds and also we can draw for coordination compounds.
Complete step by step answer:
- In the question it is given that to draw the Lewis dot structures of the beryllium difluoride.
- We know that the atomic number of beryllium is 4 and has four electrons in its electronic configuration.
- The electronic configuration of beryllium is $1{{s}^{2}}2{{s}^{2}}$
- So beryllium has two valence electrons in 2s orbital.
- The Lewis dot structure of the beryllium is as follows.
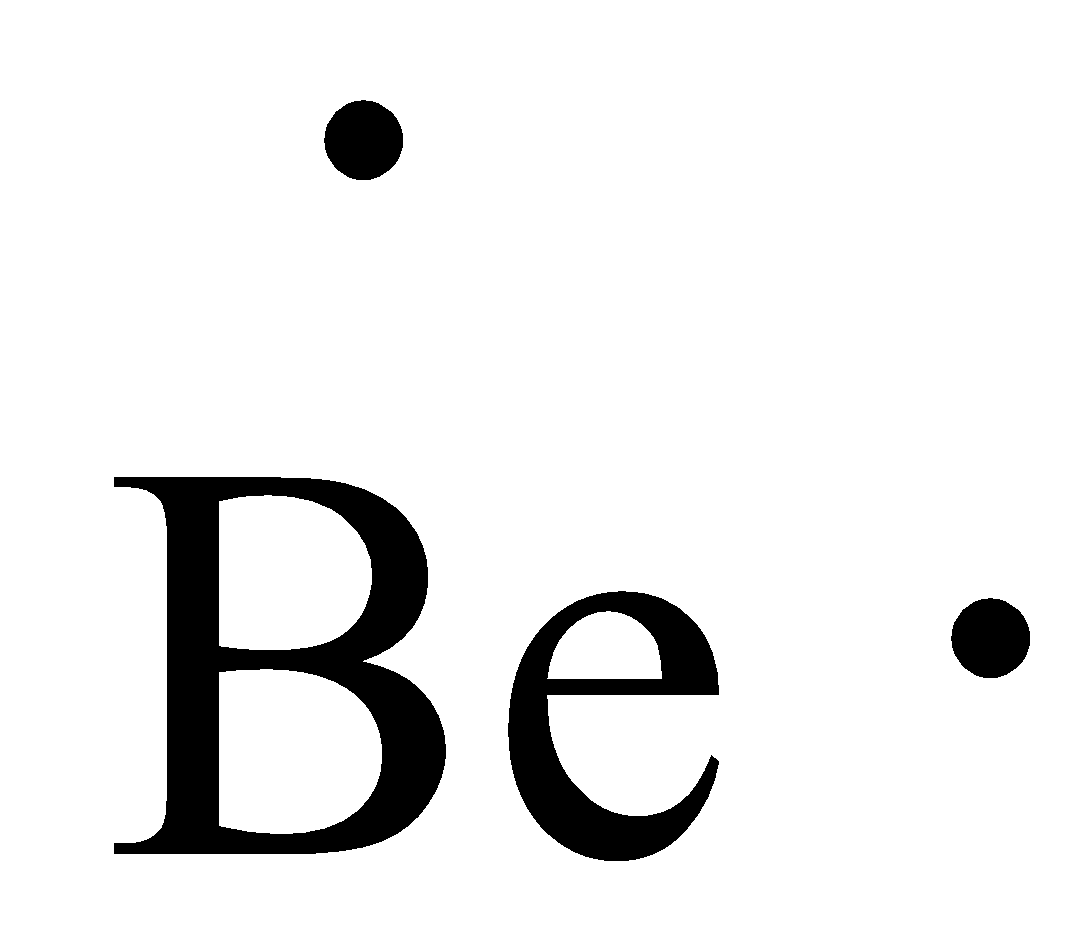
- Coming to fluorine, we know that the fluorine element has seven valence electrons in its electronic configuration.
- The Lewis dot structure of Fluorine is as follows.
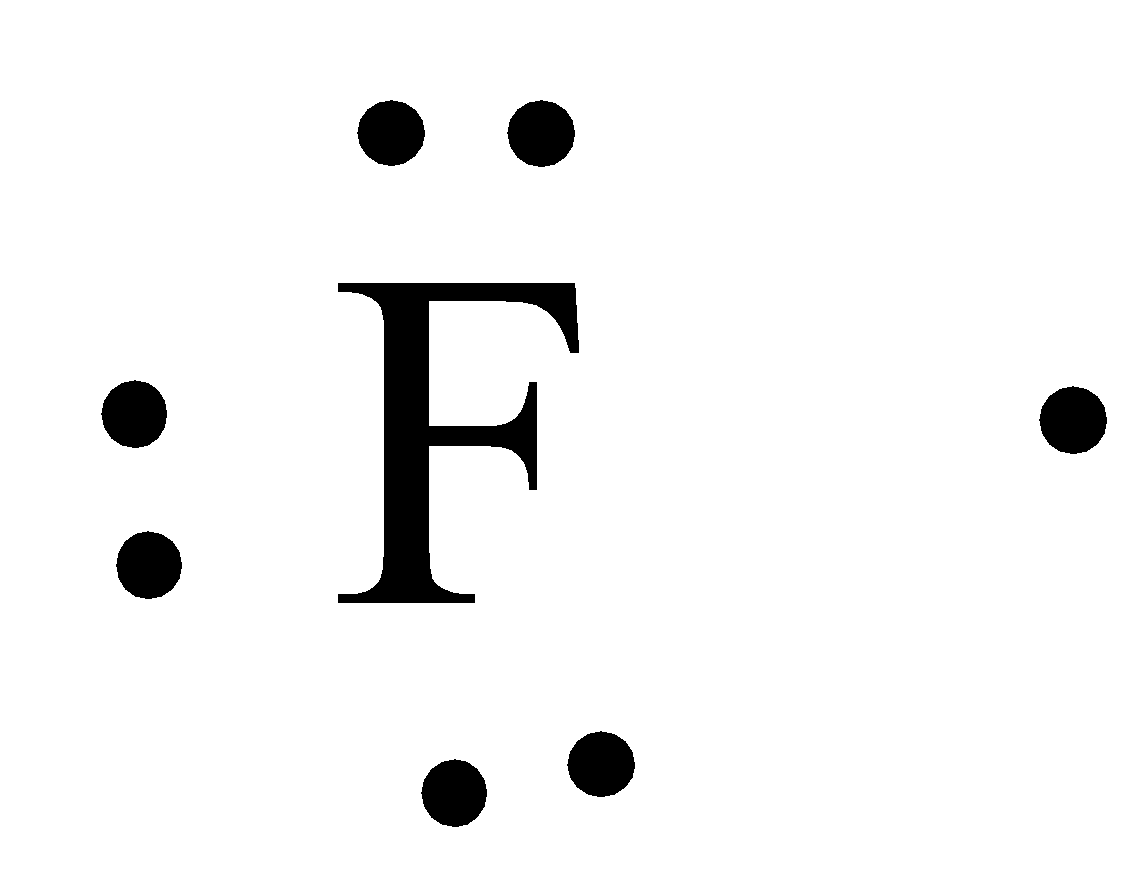
- Now by taking the above structures as examples we can draw the Lewis dot structure of $Be{{F}_{2}}$ and it is as follows.
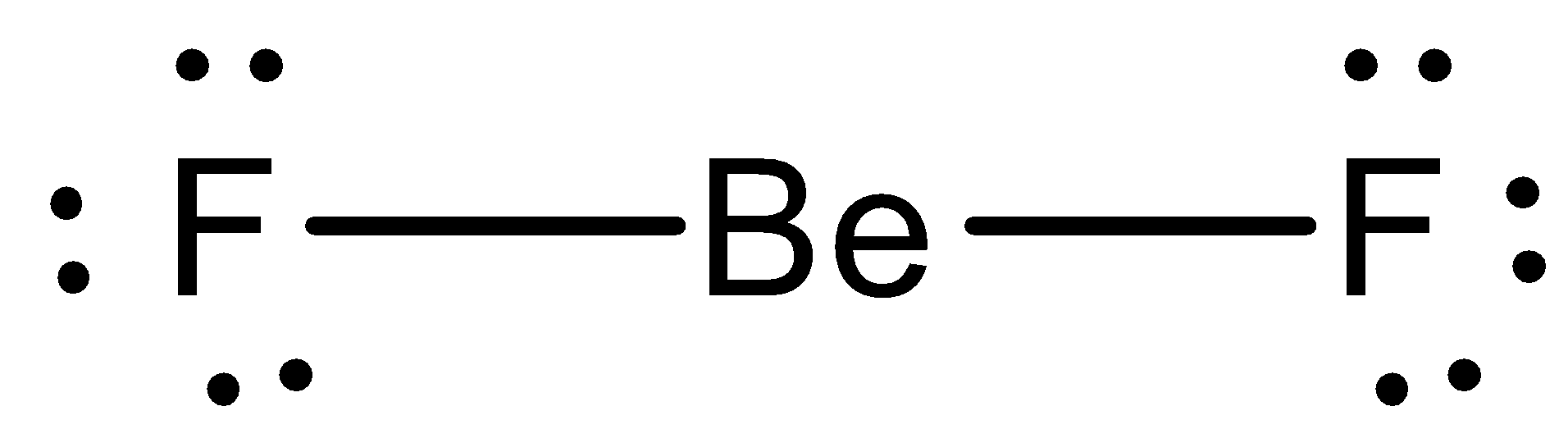
- From the above Lewis dot structure we can easily say that each fluorine atom has three lone pairs of electrons and beryllium has zero lone pair of electrons in Beryllium difluoride molecules.
Note: By using Lewis dot structures of the molecules we can determine the formation of the chemical bonds in various molecules. We can draw the Lewis dot structures for the compounds containing covalent bonds and also we can draw for coordination compounds.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




