
Draw the structure of acetonitrile.
Answer
565.5k+ views
Hint:. Acetonitrile is also known as methyl cyanide. It is a chemical compound with the molecular formula \[C{H_3}C \equiv N\]. We know that carbon will be the central metal atom and Nitrogen and the other carbon atom with methyl group will be attached to it.
Complete step by step answer:
Let’s first see what are the elements present in the acetonitrile. Acetonitrile is actually the IUPAC name for methyl cyanide. Acetonitrile is having a formula of \[C{H_3}C \equiv N\]. From the formula we can see that there are three elements present in the carbon atom. They are C, N and H atoms. There are two carbon atoms present in the acetonitrile.
- Carbon will be the central atom in this compound. Carbon is having a valency of 4. It can form only four bonds, whereas nitrogen will have a valency of 3. Since nitrogen is not bonded to any other atom, all the three-valence electrons will be forming a triple bond with the three-valence electron of the carbon atom. Remaining there will be one valence electron in the central carbon atom, it will be forming a single bond with the other carbon atom. Now the other carbon is left with three valence electrons. These electrons will be forming a single bond with three hydrogen with one valence electron. This leads to the formation of methyl groups.
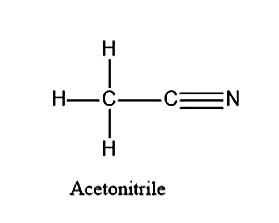
Additional information: Acetonitrile have the following characteristics.
- Acetonitrile is a transparent liquid.
- It has an aromatic odour.
- It is a polar aprotic solvent.
- There are other names for acetonitrile, they are ethyl nitrile, methyl cyanide and ethane nitrile.
- Acetonitrile has various applications such as, it is used in perfumes, pharmaceuticals, nail polish removers, pesticides and rubber products.
Note: The carbon which is attached to the nitrogen group will have \[sp\] hybridization because it is attached to only two atoms namely nitrogen and the carbon of the methyl group. The methyl group carbon will be having a hybridization of \[s{p^3}\] because it has been attached to 4 atoms namely, 3 hydrogen atoms and one carbon atom.
Complete step by step answer:
Let’s first see what are the elements present in the acetonitrile. Acetonitrile is actually the IUPAC name for methyl cyanide. Acetonitrile is having a formula of \[C{H_3}C \equiv N\]. From the formula we can see that there are three elements present in the carbon atom. They are C, N and H atoms. There are two carbon atoms present in the acetonitrile.
- Carbon will be the central atom in this compound. Carbon is having a valency of 4. It can form only four bonds, whereas nitrogen will have a valency of 3. Since nitrogen is not bonded to any other atom, all the three-valence electrons will be forming a triple bond with the three-valence electron of the carbon atom. Remaining there will be one valence electron in the central carbon atom, it will be forming a single bond with the other carbon atom. Now the other carbon is left with three valence electrons. These electrons will be forming a single bond with three hydrogen with one valence electron. This leads to the formation of methyl groups.

Additional information: Acetonitrile have the following characteristics.
- Acetonitrile is a transparent liquid.
- It has an aromatic odour.
- It is a polar aprotic solvent.
- There are other names for acetonitrile, they are ethyl nitrile, methyl cyanide and ethane nitrile.
- Acetonitrile has various applications such as, it is used in perfumes, pharmaceuticals, nail polish removers, pesticides and rubber products.
Note: The carbon which is attached to the nitrogen group will have \[sp\] hybridization because it is attached to only two atoms namely nitrogen and the carbon of the methyl group. The methyl group carbon will be having a hybridization of \[s{p^3}\] because it has been attached to 4 atoms namely, 3 hydrogen atoms and one carbon atom.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

What is a transformer Explain the principle construction class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE




