
Formation of oxonium salts shows that ethers are:
A.Basic in nature
B.Acidic in nature
C.Neutral in nature
D.Amphoteric in nature
Answer
571.5k+ views
Hint: First of all we will consider what are oxonium salts and the reaction involved in their formation. For the preliminary knowledge oxonium salts are formed when ether is reacted with a strong acid.
Complete step by step answer:
Oxonium salts are formed when ethers react with mineral acids such as hydrochloric acid or sulphuric acid. In ethers due to the presence of lone pairs of electrons on oxygen atoms, it behaves as a Lewis base and reacts with a strong acid to form protonated oxonium salt.
As oxygen atoms donate the lone pair of electrons therefore, the ether can be said to be basic in nature.
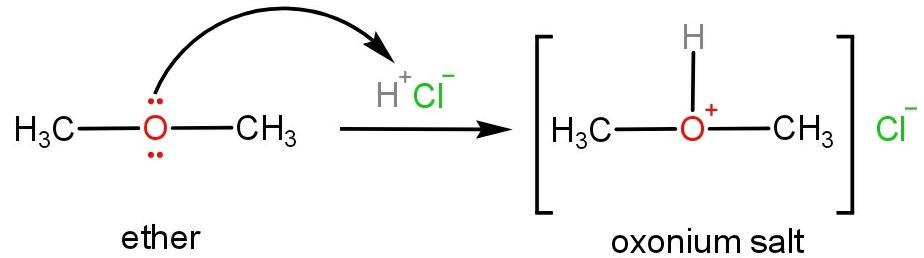
Molecular, atomic or ionic species which have a tendency to donate lone pairs of electrons or accept protons are said to be basic in nature. While species which tend to accept lone pairs of electrons or donate protons are said to be acidic in nature.
Some species act as both acid and base under different conditions. Such species which have both acidic and basic nature are called amphoteric.
Therefore, the answer is option A.
Note:
Students should not misinterpret that the reaction with a strong acid will lead to dehydration in the compound and form a dehydrated product. They should emphasize on the fact that due to the lone pair of electrons on oxygen atoms, it will tend to attract protons dissociated by the strong acid and form salt.
Complete step by step answer:
Oxonium salts are formed when ethers react with mineral acids such as hydrochloric acid or sulphuric acid. In ethers due to the presence of lone pairs of electrons on oxygen atoms, it behaves as a Lewis base and reacts with a strong acid to form protonated oxonium salt.
As oxygen atoms donate the lone pair of electrons therefore, the ether can be said to be basic in nature.

Molecular, atomic or ionic species which have a tendency to donate lone pairs of electrons or accept protons are said to be basic in nature. While species which tend to accept lone pairs of electrons or donate protons are said to be acidic in nature.
Some species act as both acid and base under different conditions. Such species which have both acidic and basic nature are called amphoteric.
Therefore, the answer is option A.
Note:
Students should not misinterpret that the reaction with a strong acid will lead to dehydration in the compound and form a dehydrated product. They should emphasize on the fact that due to the lone pair of electrons on oxygen atoms, it will tend to attract protons dissociated by the strong acid and form salt.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




