
In ammonia, the bond angle of $H-N-H$ is ______.
A. $106.7{}^\circ $
B. $104.5{}^\circ $
C. $109.5{}^\circ $
D. $120{}^\circ $
Answer
233.1k+ views
Hint: Think about the geometry of the ammonia molecule according to the hybridization of the nitrogen atom. Consider the standard angle known for that type of geometry and then take into consideration the number of lone pairs and bond pairs present.
Complete step by step solution:
We know that the number of valence electrons in the nitrogen atom is 7. The electronic configuration of nitrogen is $1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}$. So, the ground state and hybridized state of the nitrogen atom will be:
- Ground state
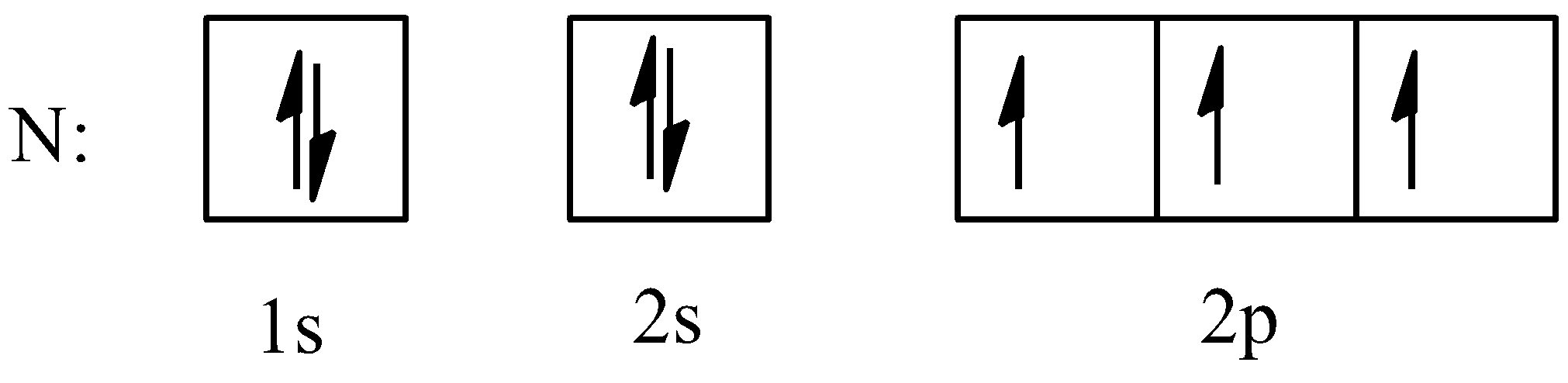
- Hybridized state
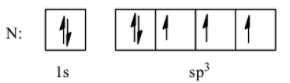
So, here we can see that the second orbital undergoes $s{{p}^{3}}$ hybridization and forms four degenerate (having same energy) orbitals. One of those orbitals will be occupied by a lone pair and the other three will be occupied by bond pairs of electrons. We know that the atoms that undergo $s{{p}^{3}}$ hybridization have a tetrahedral geometry, and the standard angle for tetrahedral geometry is $109.5{}^\circ $. But here, we need to consider the fact that not all the orbitals are occupied by bond pair interactions. One of the orbitals is occupied by the lone pair. We know that the repulsion between two lone pairs is the most followed by the repulsion between a lone pair and a bond pair and the repulsion between two bond pairs is the least. So, $\text{lone pair-lone pair}>\text{lone pair-bond pair}>\text{bond pair-bond pair}$.
The standard angle between two orbitals in tetrahedral geometry will be reduced due to the presence of this lone pair. The angle will be less than $109.5{}^\circ $. The structure of the ammonia molecule will be:

Here, we can see that the angle of the bond $H-N-H$ is less than the standard angle.
Hence, the answer to this question is ‘A. $106.7{}^\circ $’.
Note: Remember that the methane molecule has the standard tetrahedral geometry so it will exhibit the standard angle, the presence of a lone pair changes the angle in case of ammonia. Considering the water molecule too, oxygen has 2 lone pairs so the angle between $H-O-H$ will be even lesser than $106.7{}^\circ $.
Complete step by step solution:
We know that the number of valence electrons in the nitrogen atom is 7. The electronic configuration of nitrogen is $1{{s}^{2}}2{{s}^{2}}2{{p}^{3}}$. So, the ground state and hybridized state of the nitrogen atom will be:
- Ground state

- Hybridized state

So, here we can see that the second orbital undergoes $s{{p}^{3}}$ hybridization and forms four degenerate (having same energy) orbitals. One of those orbitals will be occupied by a lone pair and the other three will be occupied by bond pairs of electrons. We know that the atoms that undergo $s{{p}^{3}}$ hybridization have a tetrahedral geometry, and the standard angle for tetrahedral geometry is $109.5{}^\circ $. But here, we need to consider the fact that not all the orbitals are occupied by bond pair interactions. One of the orbitals is occupied by the lone pair. We know that the repulsion between two lone pairs is the most followed by the repulsion between a lone pair and a bond pair and the repulsion between two bond pairs is the least. So, $\text{lone pair-lone pair}>\text{lone pair-bond pair}>\text{bond pair-bond pair}$.
The standard angle between two orbitals in tetrahedral geometry will be reduced due to the presence of this lone pair. The angle will be less than $109.5{}^\circ $. The structure of the ammonia molecule will be:

Here, we can see that the angle of the bond $H-N-H$ is less than the standard angle.
Hence, the answer to this question is ‘A. $106.7{}^\circ $’.
Note: Remember that the methane molecule has the standard tetrahedral geometry so it will exhibit the standard angle, the presence of a lone pair changes the angle in case of ammonia. Considering the water molecule too, oxygen has 2 lone pairs so the angle between $H-O-H$ will be even lesser than $106.7{}^\circ $.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

Hydrocarbons Class 11 Chemistry Chapter 9 CBSE Notes - 2025-26

Thermodynamics Class 11 Chemistry Chapter 5 CBSE Notes - 2025-26

Equilibrium Class 11 Chemistry Chapter 6 CBSE Notes - 2025-26

Organic Chemistry Some Basic Principles And Techniques Class 11 Chemistry Chapter 8 CBSE Notes - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 7 Redox Reactions (2025-26)




