
In $ICl_4^ - $ , the shape is square planar, the number of bond pair-lone pair repulsion at ${90^ \circ }$ are:
$A.$ $6$
$B.$ $8$
$C.$ $12$
$D.$ $4$
Answer
543.6k+ views
Hint:We can predict the number of bond pair-lone pair repulsions at${90^ \circ }$ by drawing its structure. The shape of $ICl_4^ - $ is square planar because it has four bond pairs and lone pairs. The geometry is octahedral.
Complete step-by-step answer:Before solving the question we have to look at the structure to determine their number of bond pair-lone pairs.
Now, the structure of $ICl_4^ - $ is,
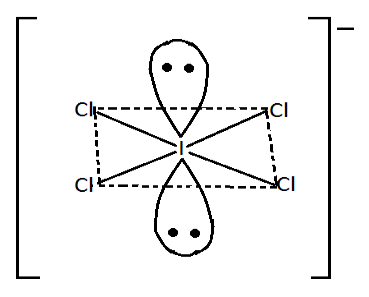
Iodine $\left( I \right)$ has $7$ electron it its valence shell in $ICl_4^ - $eight electron participate in formation of compound
Shape of $ICl_4^ - $is square planar. Since its geometry is octahedral, lone pairs reside above and below the square planar. In $ICl_4^ - $each lone pair interacts with the $4$ bond pair.
So, Number of bond pair-lone pair repulsion=Total number of lone pairs multiplied by number of bond pairs by which they interact. We have two lone pairs and four bond pairs
Number of bond pairs-lone pairs repulsion$ = 2 \times 4$
$ = 8$
Thus, there is $8$ bond pairs-lone pairs repulsion at ${90^ \circ }$ . So, the correct option is $B.$
Note: Valence Shell Electron Pair Repulsion Theory $\left( {VSEPR} \right)$ .This is a very useful theory to predict the geometry or shape of a number of polyatomic molecules or ions on a non-transition element. This theory says that shapes of a species depend on the number of and nature of electron pairs surrounding the central atom of a species.
Table summarizes the relationship between the number of electron pairs that are bond pairs and lone pairs and the shape or geometry.
Complete step-by-step answer:Before solving the question we have to look at the structure to determine their number of bond pair-lone pairs.
Now, the structure of $ICl_4^ - $ is,

Iodine $\left( I \right)$ has $7$ electron it its valence shell in $ICl_4^ - $eight electron participate in formation of compound
Shape of $ICl_4^ - $is square planar. Since its geometry is octahedral, lone pairs reside above and below the square planar. In $ICl_4^ - $each lone pair interacts with the $4$ bond pair.
So, Number of bond pair-lone pair repulsion=Total number of lone pairs multiplied by number of bond pairs by which they interact. We have two lone pairs and four bond pairs
Number of bond pairs-lone pairs repulsion$ = 2 \times 4$
$ = 8$
Thus, there is $8$ bond pairs-lone pairs repulsion at ${90^ \circ }$ . So, the correct option is $B.$
Note: Valence Shell Electron Pair Repulsion Theory $\left( {VSEPR} \right)$ .This is a very useful theory to predict the geometry or shape of a number of polyatomic molecules or ions on a non-transition element. This theory says that shapes of a species depend on the number of and nature of electron pairs surrounding the central atom of a species.
Table summarizes the relationship between the number of electron pairs that are bond pairs and lone pairs and the shape or geometry.
| Total number of electron pairs | Shape |
| 2 | Linear |
| 3 | Trigonal planar |
| 4 | Tetrahedral |
| 5 | Trigonal bipyramidal |
| 6 | Octahedral |
| 7 | Pentagonal bipyramidal |
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




