
Number of Sigma and Pi bonds in $Cl{O_2}^ - $ ion.
A. 2 $\sigma $and 2 $\pi $
B. 2 $\sigma $and 1 $\pi $
C. 1 $\sigma $ and 2 $\pi $
D. 3 $\sigma $ and 2 $\pi $
Answer
559.8k+ views
Hint:firstly in this type of question we make the bonds structure of $Cl{O_2}^ - $ ion means that we will make molecular structure of the given compound $Cl{O_2}^ - $ after this we will calculate the number of $\sigma $ bonds and the $\pi $ bonds if we have one Bond then it will be $\sigma $ and if will have two bonds then it will be one $\sigma $ and one $\pi $ and if we have three bonds means it will be one Sigma and two $\pi $ bonds.
Complete step-by-step answer:firstly in this type of question we make the bonds structure of $Cl{O_2}^ - $ ion means that we will make molecular structure of the given compound $Cl{O_2}^ - $ after this we will calculate the number of $\sigma $ bonds and the $\pi $ bonds if we have one Bond then it will be $\sigma $ and if will have two bonds then it will be one $\sigma $ and one $\pi $ and if we have three bonds means it will be one Sigma and two $\pi $ bonds.
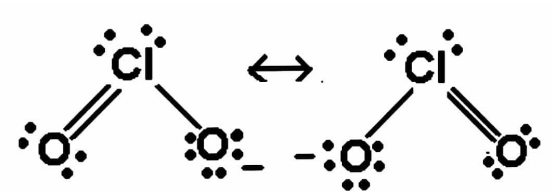
The above shown structure is the molecular structure of the given ion.
We can form the resonance structure of a given compound but there will be no change in the sigma bond and the pi bond of that ion.
Now we can see in the figure that there are 2 bonds in one oxygen atom and 1 bond in one oxygen atom.
$\therefore $we have already discussed that two bonds means one sigma and one pi. and one bond means one sigma bond
Hence, we can say that in the given compound we have 2 sigma bonds and one pi bond.
Therefore, the correct answer will be option number B.
Note:As we all know that $\sigma $ bond is formed when overlapping orbitals will be head to head and Pi bonds are formed when orbitals overlap side by side. The Sigma bond is much stronger than Pi bonds in any compound.
Resonance structure: multiple structures of a compound showing the movement of electrons in a molecular structure is known as resonance and the number of structures of that compound is called resonance structure.
Complete step-by-step answer:firstly in this type of question we make the bonds structure of $Cl{O_2}^ - $ ion means that we will make molecular structure of the given compound $Cl{O_2}^ - $ after this we will calculate the number of $\sigma $ bonds and the $\pi $ bonds if we have one Bond then it will be $\sigma $ and if will have two bonds then it will be one $\sigma $ and one $\pi $ and if we have three bonds means it will be one Sigma and two $\pi $ bonds.

The above shown structure is the molecular structure of the given ion.
We can form the resonance structure of a given compound but there will be no change in the sigma bond and the pi bond of that ion.
Now we can see in the figure that there are 2 bonds in one oxygen atom and 1 bond in one oxygen atom.
$\therefore $we have already discussed that two bonds means one sigma and one pi. and one bond means one sigma bond
Hence, we can say that in the given compound we have 2 sigma bonds and one pi bond.
Therefore, the correct answer will be option number B.
Note:As we all know that $\sigma $ bond is formed when overlapping orbitals will be head to head and Pi bonds are formed when orbitals overlap side by side. The Sigma bond is much stronger than Pi bonds in any compound.
Resonance structure: multiple structures of a compound showing the movement of electrons in a molecular structure is known as resonance and the number of structures of that compound is called resonance structure.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

10 examples of friction in our daily life




