
Outline the reaction sequence for the conversion of ethene to ethyne (the number of steps should not be more than two).
A. A:$\text{HOCl}$ B: $\text{KOH}$
B. A: bromine water B: Diethylamine
C. A:$\text{C}{{\text{l}}_{2}}\text{,hv}$ B:$\text{Ca(OH}{{\text{)}}_{2}}$
D. A: bromine water B: $\text{KOH}$
Answer
593.7k+ views
Hint: We have to perform mechanisms or perform the steps by the reagents given to achieve our product. Ethene has two carbon atoms with a double bond and rest vacancies are filled by hydrogen atoms. Ethyne has two carbon atoms with a triple bond and rest vacancies are filled by hydrogen atoms.
Complete answer:
The question can be directly solved as the conversion of ethene to ethyne is given when we talk about preparation of ethynes. Let us see that reaction and the steps to convert ethene to Ethyne are:
(1) Ethene has a chemical formula $\text{C}{{\text{H}}_{2}}\text{=C}{{\text{H}}_{2}}$.
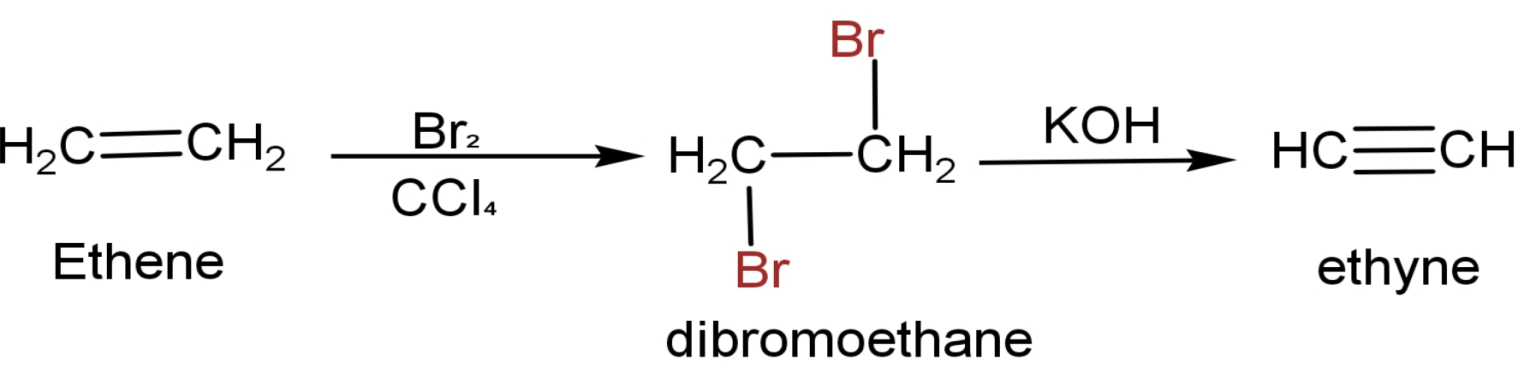
(2) Made it react with $\text{B}{{\text{r}}_{2}}$ in the non-polar solvent $\text{CC}{{\text{l}}_{4}}$.Two atoms of $\text{Br}$ attaches to both the doubled-bonded carbon and forms dibromoethane $\text{(C}{{\text{H}}_{2}}\text{Br-C}{{\text{H}}_{2}}\text{Br}$). This a type of addition reaction.
(3) Then it is heated in the presence of alcoholic $\text{KOH}$.The end product is ethyne.
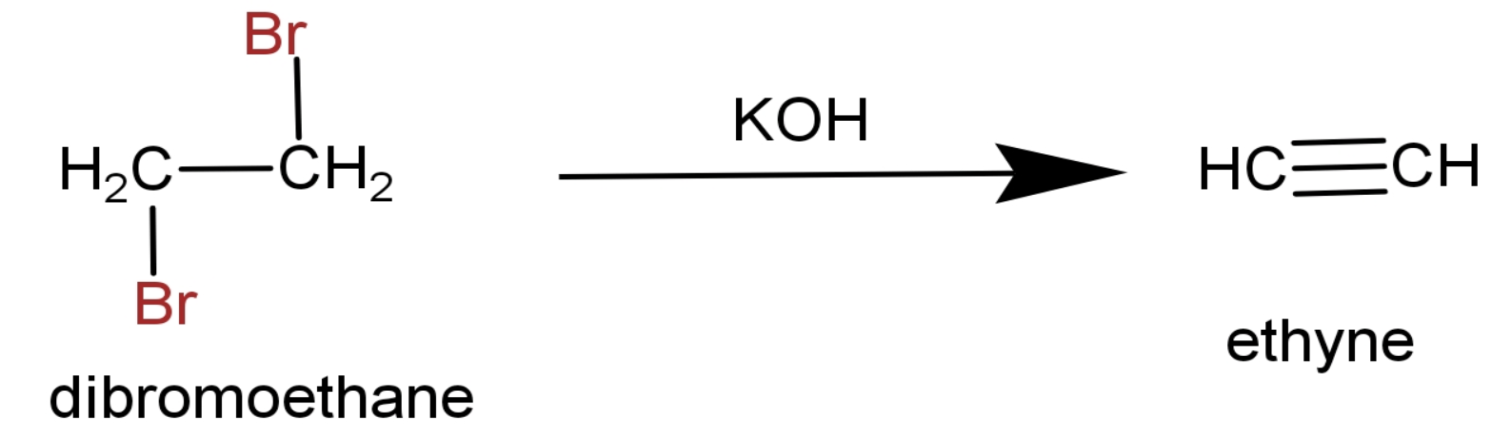
The full reaction is
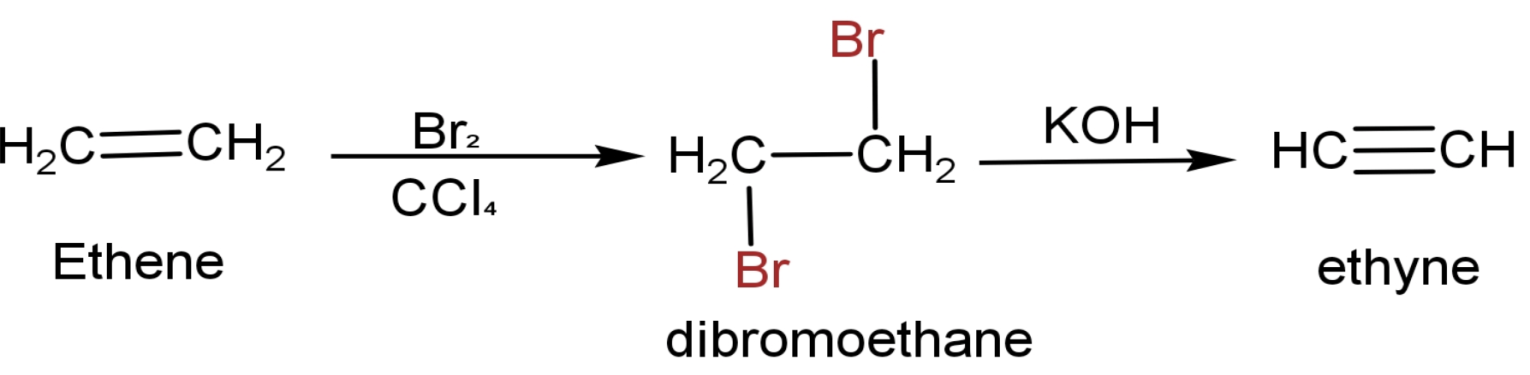
The correct answer to this question is option ‘d’ which is first bromine water then $\text{KOH}$.
So, the correct answer is “Option D”.
Additional Information: Uses of ethyne:
(1) Calcium carbide was used to produce ethyne used in the lamps for portable and remote applications.
(2) Oxy-acetylene welding equipment is versatile because a torch is preferred for iron or steel welding.
Note: The work each reagent should be known correctly. Here$\text{B}{{\text{r}}_{2}}$ is used with non-polar solvent like $\text{CC}{{\text{l}}_{4}}$ that is so that $\text{B}{{\text{r}}_{2}}$ does not react with polar solvent (like water) to form other products and reaction does not suffer.
Complete answer:
The question can be directly solved as the conversion of ethene to ethyne is given when we talk about preparation of ethynes. Let us see that reaction and the steps to convert ethene to Ethyne are:
(1) Ethene has a chemical formula $\text{C}{{\text{H}}_{2}}\text{=C}{{\text{H}}_{2}}$.

(2) Made it react with $\text{B}{{\text{r}}_{2}}$ in the non-polar solvent $\text{CC}{{\text{l}}_{4}}$.Two atoms of $\text{Br}$ attaches to both the doubled-bonded carbon and forms dibromoethane $\text{(C}{{\text{H}}_{2}}\text{Br-C}{{\text{H}}_{2}}\text{Br}$). This a type of addition reaction.
(3) Then it is heated in the presence of alcoholic $\text{KOH}$.The end product is ethyne.

The full reaction is

The correct answer to this question is option ‘d’ which is first bromine water then $\text{KOH}$.
So, the correct answer is “Option D”.
Additional Information: Uses of ethyne:
(1) Calcium carbide was used to produce ethyne used in the lamps for portable and remote applications.
(2) Oxy-acetylene welding equipment is versatile because a torch is preferred for iron or steel welding.
Note: The work each reagent should be known correctly. Here$\text{B}{{\text{r}}_{2}}$ is used with non-polar solvent like $\text{CC}{{\text{l}}_{4}}$ that is so that $\text{B}{{\text{r}}_{2}}$ does not react with polar solvent (like water) to form other products and reaction does not suffer.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




