
What is the oxidation number of Mn in $KMn{{O}_{4}}$ and of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$?
Answer
589.2k+ views
Hint: Oxidation number or oxidation state is the charge obtained by the atom of an element when it loses or gains electrons while going from its free state to combined state with atoms of other elements. It can be positive, negative or zero.
Complete answer:
> Oxidation number of alkali metals (e.g. Na, K, Li, etc) is always taken as +1.
> Oxidation number of oxygen is -2 in all of its compounds with a few exceptions. Oxidation state of O in peroxides (e.g. $N{{a}_{2}}{{O}_{2}}$, ${{H}_{2}}{{O}_{2}}$), superoxide (e.g. $K{{O}_{2}}$) and $O{{F}_{2}}$ are -1, $-\dfrac{1}{2}$ and +2, respectively.
Oxidation number of Mn in potassium permanganate, $KMn{{O}_{4}}$ is calculated as follows:
Let the oxidation number of Mn be ‘’. Oxidation of K is +1 and of O is -2. $KMn{{O}_{4}}$ is a neutral compound, so the total charge on it is taken as zero.
1(+1)+x+4(-2)=0
1+x-8=0
x=7
Hence, the oxidation number of Mn in is +7.
Oxidation number of S in sodium thiosulphate, $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$ can be calculate as:
To calculate the oxidation of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$, we have to take a look at its structure first.
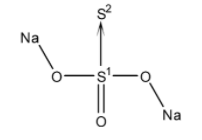
It has two sulphur atoms in different oxidation states. Since, sulphur ${{S}_{2}}$ is bonded to sulphur ${{S}_{1}}$ by a coordination double bond. ${{S}_{1}}$ is known to donate its electron pair to ${{S}_{2}}$. Therefore, the oxidation state of ${{S}_{2}}$ is -2.
Now let the oxidation number of sulphur ${{S}_{1}}$ be ‘$x$’. Both the Na atoms have +1 oxidation number and all the three O atoms are in -2 oxidation state. Total charge on the compound is 0. Thus, the oxidation number of the other sulphur atom can be calculated as:
2(+1)+x+1(-2)+3(-2)=0
2+x-2-6=0
x-6=0
x=6
Therefore, the oxidation number of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$ is +6.
Hence, the oxidation number of Mn in $KMn{{O}_{4}}$ is +7 and of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$ is +6.
Note: We commonly make an error while calculating the oxidation number of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$. We may simply calculate it to be +2 as shown below, which is wrong.
2+2x+3(-2)=0
2x-4=0
x=2
The two sulphur atoms are present in different oxidation states in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$.
Complete answer:
> Oxidation number of alkali metals (e.g. Na, K, Li, etc) is always taken as +1.
> Oxidation number of oxygen is -2 in all of its compounds with a few exceptions. Oxidation state of O in peroxides (e.g. $N{{a}_{2}}{{O}_{2}}$, ${{H}_{2}}{{O}_{2}}$), superoxide (e.g. $K{{O}_{2}}$) and $O{{F}_{2}}$ are -1, $-\dfrac{1}{2}$ and +2, respectively.
Oxidation number of Mn in potassium permanganate, $KMn{{O}_{4}}$ is calculated as follows:
Let the oxidation number of Mn be ‘’. Oxidation of K is +1 and of O is -2. $KMn{{O}_{4}}$ is a neutral compound, so the total charge on it is taken as zero.
1(+1)+x+4(-2)=0
1+x-8=0
x=7
Hence, the oxidation number of Mn in is +7.
Oxidation number of S in sodium thiosulphate, $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$ can be calculate as:
To calculate the oxidation of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$, we have to take a look at its structure first.

It has two sulphur atoms in different oxidation states. Since, sulphur ${{S}_{2}}$ is bonded to sulphur ${{S}_{1}}$ by a coordination double bond. ${{S}_{1}}$ is known to donate its electron pair to ${{S}_{2}}$. Therefore, the oxidation state of ${{S}_{2}}$ is -2.
Now let the oxidation number of sulphur ${{S}_{1}}$ be ‘$x$’. Both the Na atoms have +1 oxidation number and all the three O atoms are in -2 oxidation state. Total charge on the compound is 0. Thus, the oxidation number of the other sulphur atom can be calculated as:
2(+1)+x+1(-2)+3(-2)=0
2+x-2-6=0
x-6=0
x=6
Therefore, the oxidation number of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$ is +6.
Hence, the oxidation number of Mn in $KMn{{O}_{4}}$ is +7 and of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$ is +6.
Note: We commonly make an error while calculating the oxidation number of S in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$. We may simply calculate it to be +2 as shown below, which is wrong.
2+2x+3(-2)=0
2x-4=0
x=2
The two sulphur atoms are present in different oxidation states in $N{{a}_{2}}{{S}_{2}}{{O}_{3}}$.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

10 examples of friction in our daily life




