
How will you prepare chlorobenzene from benzene diazonium chloride?
Answer
589.5k+ views
Hint: We know that when a diazonium salt is treated with copper (I)chloride $\left( {C{u_2}C{l_2}} \right)$ or copper (I) bromide $\left( {C{u_2}B{r_2}} \right)$, the corresponding haloarene is formed. This reaction is known as the Sandmeyer reaction. It is used for introducing a chloro or bromo group in the benzene ring.
Complete step by step answer:
According to the question we have to state how chlorobenzene will be prepared from benzene diazonium chloride by the Sandmeyer reaction.
Hence , The Sandmeyer reaction is a chemical reaction used to synthesize aryl halides from aryl diazonium salts. This reaction is named after the Swiss chemist Traugott Sandmeyer.
The Sandmeyer reaction is a method for substitution of an aromatic amino group by preparation of its diazonium salt followed by its displacement with a nucleophile, often catalyzed by copper (I) salts. The nucleophile can include halide anions, cyanide , thiols, water and others.
The substitution of an aromatic amino group is possible by preparation of its diazonium salt and successively displacement with a nucleophile. General reaction equation of formation of diazonium salt and haloarenes given below:
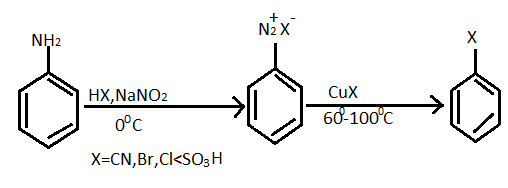
For the given question, reaction of preparation of chlorobenzene from diazonium chloride as follows :
Note:Benzene diazonium salt is formed by treating an aromatic primary amine with $NaN{O_2}$ and dil. $HCl$ at low temperature. The process is known as diazotization. Diazonium salts are highly reactive compounds. They are used in the preparation of a large number of arene derivatives. The Sandmeyer reaction helps us to provide a method through which we can perform unique transformations on benzene for example halogenation, cyanation , sulphonation and hydroxylation.
Complete step by step answer:
According to the question we have to state how chlorobenzene will be prepared from benzene diazonium chloride by the Sandmeyer reaction.
Hence , The Sandmeyer reaction is a chemical reaction used to synthesize aryl halides from aryl diazonium salts. This reaction is named after the Swiss chemist Traugott Sandmeyer.
The Sandmeyer reaction is a method for substitution of an aromatic amino group by preparation of its diazonium salt followed by its displacement with a nucleophile, often catalyzed by copper (I) salts. The nucleophile can include halide anions, cyanide , thiols, water and others.
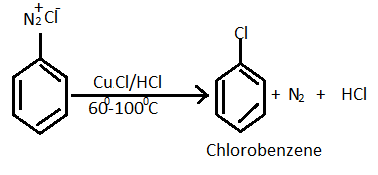
The substitution of an aromatic amino group is possible by preparation of its diazonium salt and successively displacement with a nucleophile. General reaction equation of formation of diazonium salt and haloarenes given below:

For the given question, reaction of preparation of chlorobenzene from diazonium chloride as follows :

Note:Benzene diazonium salt is formed by treating an aromatic primary amine with $NaN{O_2}$ and dil. $HCl$ at low temperature. The process is known as diazotization. Diazonium salts are highly reactive compounds. They are used in the preparation of a large number of arene derivatives. The Sandmeyer reaction helps us to provide a method through which we can perform unique transformations on benzene for example halogenation, cyanation , sulphonation and hydroxylation.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




