
What is the production of hydrogenation of trans-2-butene?
Answer
524.7k+ views
Hint :In hydrogenation, double and triple bonds get broken to single bonds. Thus, the double bond in trans-2-butene gets reduced to a single bond.
Complete Step By Step Answer:
The product of hydrogenation of trans-2-butene is simple butane.
Hydrogenation is a reaction in which triple bonds get reduced to double bonds, and double bonds get reduced to single bonds. It is a chemical reaction in which molecular hydrogen and any other compound are subjected to a chemical reaction under the presence of a catalyst such as nickel, palladium or platinum.
Now, there is a double bond in the given compound i.e. trans-2-butene. As hydrogenation reduces the double bond to single, here also the same will happen.
The below given figure is of trans-2-butene.
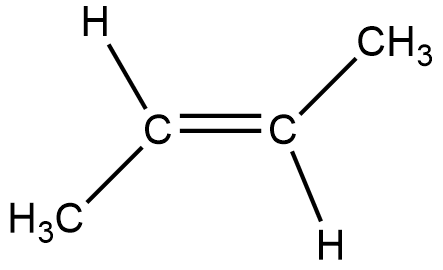
Now, after hydrogenation the double bond gets broken into a single bond, and thus both the carbon atoms get an extra hydrogen to comply with the reduced double bond.
The below given is the product after hydrogenation, which is just simple butane.

Thus, if any element or compound is subjected to a chemical reaction with molecular hydrogen, it undergoes hydrogenation and loses its triple bond to double bond and its double bond to single bond. Double hydrogenation on triple bonds can make it a single bond.
Note :
Any compound can reduce its double or triple bond to single through hydrogenation. There are other methods too, but this method is more reliable on most of the compounds.
Complete Step By Step Answer:
The product of hydrogenation of trans-2-butene is simple butane.
Hydrogenation is a reaction in which triple bonds get reduced to double bonds, and double bonds get reduced to single bonds. It is a chemical reaction in which molecular hydrogen and any other compound are subjected to a chemical reaction under the presence of a catalyst such as nickel, palladium or platinum.
Now, there is a double bond in the given compound i.e. trans-2-butene. As hydrogenation reduces the double bond to single, here also the same will happen.
The below given figure is of trans-2-butene.

Now, after hydrogenation the double bond gets broken into a single bond, and thus both the carbon atoms get an extra hydrogen to comply with the reduced double bond.
The below given is the product after hydrogenation, which is just simple butane.

Thus, if any element or compound is subjected to a chemical reaction with molecular hydrogen, it undergoes hydrogenation and loses its triple bond to double bond and its double bond to single bond. Double hydrogenation on triple bonds can make it a single bond.
Note :
Any compound can reduce its double or triple bond to single through hydrogenation. There are other methods too, but this method is more reliable on most of the compounds.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




