
The coefficient of linear expansion of a cubical crystal along three mutually perpendicular directions is ${\alpha _1}$ ${\alpha _2}$ and ${\alpha _3}$ . What is the coefficient of cubical expansion of the crystal?
Answer
471.6k+ views
Hint: You may start with a cuboid and set the temperature to rise from ${0^ \circ }C$ to ${T^ \circ }C$ as a first step. Then you might find the linear expansion equation in all three dimensions. The volume of the solid will then be determined by taking their product. You can get an expression by expanding that expression and then ignoring the smaller terms. You may figure out the answer by comparing it to the volume expansion expression.
Complete answer:
Considering a cubical crystal structure
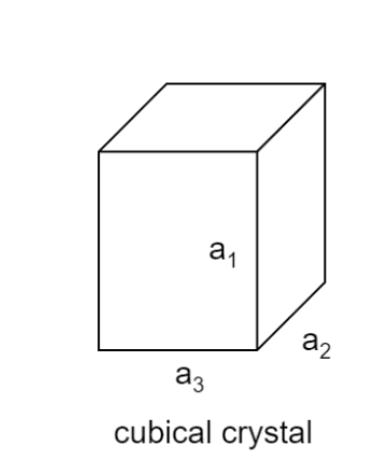
Let ${\alpha _1}$ ${\alpha _2}$ and ${\alpha _3}$ . be the coefficient of linear expression along the sides is ${a_1}$ ${a_2}$ and ${a_3}$.
We know for linear expansion
$a = {a_0}(1 + \alpha \Delta T)$
Where ${a_0}$ is the initial edge length
Take the temperature of the solid above to rise from ${0^ \circ }C$ to ${T^ \circ }C$
\[\Delta T = T - 0 = T\]
So for all the edges we have
\[{a_1}\prime = {a_1}(1 + {\alpha _1}T)\]
\[{a_2}\prime = {a_2}(1 + {\alpha _2}T)\]
\[{a_3}\prime = {a_3}(1 + {\alpha _3}T)\]
After expansion, their product will give the volume of the cuboid at temperature ${T^ \circ }C$ .
\[V\prime = {a_1}\prime {a_2}\prime {a_3}\prime = {a_1}{a_2}{a_3}(1 + {\alpha _1}T)(1 + {\alpha _2}T)(1 + {\alpha _3}T)\]
However, we know that the cuboid's initial volume is determined by the product of the edges. That is to say,
\[{V_0} = {a_1}{a_2}{a_3}\]
By neglecting small values from the above equation we get
\[V\prime \approx {V_0}(1 + ({\alpha _1} + {\alpha _2} + {\alpha _3})T)\]
Now using the formula of volume expansion
\[V = {V_0}(1 + \gamma \Delta T)\]
Comparing this from the above equation we get
\[\gamma = {\alpha _1} + {\alpha _2} + {\alpha _3}\]
Note:
Thermal expansion is the phrase used to describe the tendency of matter to change in length, area, and volume as a result of temperature changes. When molecules are heated, they move and vibrate more, increasing the space between them. The ratio of relative expansion to temperature change can be characterised as the coefficient of thermal expansion.
Complete answer:
Considering a cubical crystal structure

Let ${\alpha _1}$ ${\alpha _2}$ and ${\alpha _3}$ . be the coefficient of linear expression along the sides is ${a_1}$ ${a_2}$ and ${a_3}$.
We know for linear expansion
$a = {a_0}(1 + \alpha \Delta T)$
Where ${a_0}$ is the initial edge length
Take the temperature of the solid above to rise from ${0^ \circ }C$ to ${T^ \circ }C$
\[\Delta T = T - 0 = T\]
So for all the edges we have
\[{a_1}\prime = {a_1}(1 + {\alpha _1}T)\]
\[{a_2}\prime = {a_2}(1 + {\alpha _2}T)\]
\[{a_3}\prime = {a_3}(1 + {\alpha _3}T)\]
After expansion, their product will give the volume of the cuboid at temperature ${T^ \circ }C$ .
\[V\prime = {a_1}\prime {a_2}\prime {a_3}\prime = {a_1}{a_2}{a_3}(1 + {\alpha _1}T)(1 + {\alpha _2}T)(1 + {\alpha _3}T)\]
However, we know that the cuboid's initial volume is determined by the product of the edges. That is to say,
\[{V_0} = {a_1}{a_2}{a_3}\]
By neglecting small values from the above equation we get
\[V\prime \approx {V_0}(1 + ({\alpha _1} + {\alpha _2} + {\alpha _3})T)\]
Now using the formula of volume expansion
\[V = {V_0}(1 + \gamma \Delta T)\]
Comparing this from the above equation we get
\[\gamma = {\alpha _1} + {\alpha _2} + {\alpha _3}\]
Note:
Thermal expansion is the phrase used to describe the tendency of matter to change in length, area, and volume as a result of temperature changes. When molecules are heated, they move and vibrate more, increasing the space between them. The ratio of relative expansion to temperature change can be characterised as the coefficient of thermal expansion.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




