
The ${\text{pKa}}$ of acetyl salicylic acid (aspirin) is $3.5$ . The ${\text{pH}}$ of gastric juice in the human stomach is about $2 - 3$ and the ${\text{pH}}$ in the small intestine is about $8$ . Aspirin will be
A.Unionised in the small intestine and in the stomach
B.Completely ionised in the small intestine and in the stomach
C.Ionised in the stomach and the almost unionised in the small intestine
D.Ionised in the small intestine and almost unionised in the stomach
Answer
587.1k+ views
Hint: The ${\text{pKa}}$ value and ${\text{pH}}$ value has a unique relation in them. Basically the ${\text{pKa}}$ is the ${\text{pH}}$ value at which chemical species start accepting and donating protons. The lower the ${\text{pKa}}$ value the stronger is the acid with the ability to donate protons in aqueous solution.
Complete step by step answer:
We have given that the ${\text{pKa}}$ of acetyl salicylic acid (aspirin) is $3.5$ and the ${\text{pH}}$ of gastric juice in human stomach is about $2 - 3$ and the ${\text{pH}}$ in the small intestine is about $8$. This indicates that we have to find out the relation between ${\text{pKa}}$ value and ${\text{pH}}$ value.
And we know that the ${\text{pKa}}$ is the ${\text{pH}}$ value at which a species starts donating or accepting the protons.
Therefore we can say that the higher the ${\text{pKa}}$ value, the more the species are acidic.
The acid with higher ${\text{pKa}}$ value will be ionised in the space having higher ${\text{pH}}$ value and the acid with lower ${\text{pKa}}$ value will be ionised in the space having lower ${\text{pH}}$ value, other than their matching conditions they will remain unionised.
As we all know that aspirin is a weak acid so it will not ionise in medium with lower ${\text{pH}}$ value but it will be ionised in the medium with higher ${\text{pH}}$ .
Hence the human stomach is more acidic than the small intestine so aspirin will remain unionised in the stomach and will ionise in the small intestine.
Hence option (D) is correct.
Note:
The structure of Aspirin:
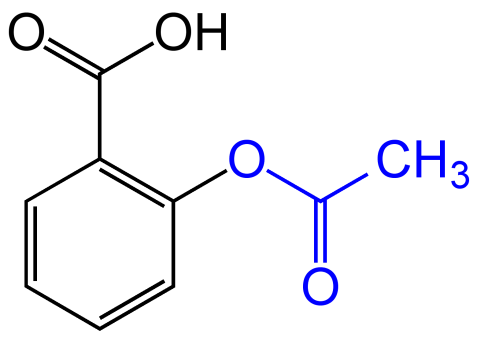
Aspirin is a weak acid therefore it has higher ${\text{pKa}}$ and ${\text{pH}}$ values and this makes it suitable to ionize in small intestine and remain unionised in the stomach.
Complete step by step answer:
We have given that the ${\text{pKa}}$ of acetyl salicylic acid (aspirin) is $3.5$ and the ${\text{pH}}$ of gastric juice in human stomach is about $2 - 3$ and the ${\text{pH}}$ in the small intestine is about $8$. This indicates that we have to find out the relation between ${\text{pKa}}$ value and ${\text{pH}}$ value.
And we know that the ${\text{pKa}}$ is the ${\text{pH}}$ value at which a species starts donating or accepting the protons.
Therefore we can say that the higher the ${\text{pKa}}$ value, the more the species are acidic.
The acid with higher ${\text{pKa}}$ value will be ionised in the space having higher ${\text{pH}}$ value and the acid with lower ${\text{pKa}}$ value will be ionised in the space having lower ${\text{pH}}$ value, other than their matching conditions they will remain unionised.
As we all know that aspirin is a weak acid so it will not ionise in medium with lower ${\text{pH}}$ value but it will be ionised in the medium with higher ${\text{pH}}$ .
Hence the human stomach is more acidic than the small intestine so aspirin will remain unionised in the stomach and will ionise in the small intestine.
Hence option (D) is correct.
Note:
The structure of Aspirin:

Aspirin is a weak acid therefore it has higher ${\text{pKa}}$ and ${\text{pH}}$ values and this makes it suitable to ionize in small intestine and remain unionised in the stomach.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

What organs are located on the left side of your body class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




