
The P-T and V-T diagrams of an isochoric process of $n$ moles of an ideal gas from pressure ${P_0}$, volume ${V_0}$ to pressure $4{P_0}$, indicating the pressures and temperatures of the gas in the initial and the final states will be __________.
Answer
568.8k+ views
Hint : In an isochoric process, the volume of the system remains constant while the system undergoes the process. Using the ideal gas law we can relate the pressure, volume, and temperature of an ideal gas in the initial and the final states of the gas.
Formula used: In this question, we will use the following formula:
$PV = nRT$where $P$ is the pressure of the gas, $V$ is the volume, $n$ is the number of moles, $R$ is the gas constant, and $T$ is the temperature of the gas
Complete step by step answer
We’ve been given an isochoric process of $n$ moles of an ideal gas from pressure ${P_0}$, volume ${V_0}$ to pressure $4{P_0}$. Since the process is isochoric, the volume of the process remains constant and we can say that the volume of the gas in the final state will also be ${V_0}$.
Using the ideal gas law, we can write
$PV = nRT$
$ \Rightarrow V = \dfrac{{nRT}}{P}$
Since the volume of the gas remains constant, we can write
$\dfrac{{{T_1}}}{{{P_1}}} = \dfrac{{{T_2}}}{{{P_2}}}$
$\dfrac{{{T_1}}}{{{P_0}}} = \dfrac{{{T_2}}}{{4{P_0}}}$
Cross multiplying the numerator and denominator, we get
$4{T_1} = {T_2}$
We can now form the P-T and V-T diagrams knowing the initial and final stage of the gas as shown below
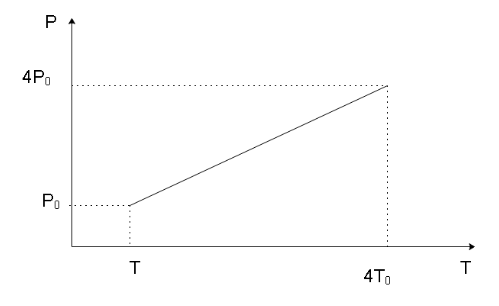
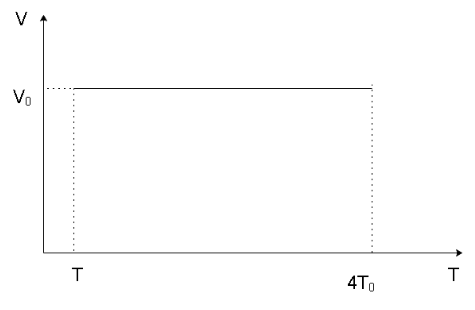
Note
Since the process is isochoric, we can get a hint that in the P-V graph, the line indicating the process will be parallel to the $y$ -axis. Similarly, in the V-T graph, the line indicating the process will be parallel to the $x$ -axis which can also be stated as the process line will be in a direction perpendicular to the direction of the volume axis as the volume of the system remains constant in the process and will have a singular value.
Formula used: In this question, we will use the following formula:
$PV = nRT$where $P$ is the pressure of the gas, $V$ is the volume, $n$ is the number of moles, $R$ is the gas constant, and $T$ is the temperature of the gas
Complete step by step answer
We’ve been given an isochoric process of $n$ moles of an ideal gas from pressure ${P_0}$, volume ${V_0}$ to pressure $4{P_0}$. Since the process is isochoric, the volume of the process remains constant and we can say that the volume of the gas in the final state will also be ${V_0}$.
Using the ideal gas law, we can write
$PV = nRT$
$ \Rightarrow V = \dfrac{{nRT}}{P}$
Since the volume of the gas remains constant, we can write
$\dfrac{{{T_1}}}{{{P_1}}} = \dfrac{{{T_2}}}{{{P_2}}}$
$\dfrac{{{T_1}}}{{{P_0}}} = \dfrac{{{T_2}}}{{4{P_0}}}$
Cross multiplying the numerator and denominator, we get
$4{T_1} = {T_2}$
We can now form the P-T and V-T diagrams knowing the initial and final stage of the gas as shown below


Note
Since the process is isochoric, we can get a hint that in the P-V graph, the line indicating the process will be parallel to the $y$ -axis. Similarly, in the V-T graph, the line indicating the process will be parallel to the $x$ -axis which can also be stated as the process line will be in a direction perpendicular to the direction of the volume axis as the volume of the system remains constant in the process and will have a singular value.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




