
To protect silk clothes and woollen clothes, we should use:
(A) Medicines
(B) Salt solution
(C) Benzene solution
(D) Naphthalene balls
Answer
587.7k+ views
Hint: Insects easily attack the silk clothes and woollen clothes, so it has to be protected with an insect repellant. The substance which is used to protect the clothes is solid and it directly converts into gas.
Complete step by step solution:
To protect silk clothes and woollen clothes we use naphthalene balls.
When the clothes are kept for a long time the insects start to ruin the material of the clothes. So, these clothes have to be protected from insects. Therefore, we use naphthalene balls.
The naphthalene balls are insect repellant, so these balls can be directly kept between the clothes. The naphthalene has a property of insect repellent because of its odour.
Naphthalene is an aromatic hydrocarbon that has a formula ${{C}_{10}}{{H}_{8}}$. The structure of naphthalene is given below:
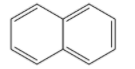
It has a concentrated odour which penetrates very easily. This odour makes it useful as an insect or moth repellent.
The naphthalene balls have the property for getting converted into gas directly from solid. This process is called sublimation. So what happens is the solid naphthalene balls kept in the clothes start to get directly converted into gas. This gas is a toxic gas, which is used to kill or repel insects. As these balls directly convert into gas, it leaves no residue.
So the correct answer is an option (D)- Naphthalene balls.
Note: Not only in clothes but also in washrooms, cupboards, etc. naphthalene balls are used for protection from insects. These naphthalene balls are toxic for both adult and larva form of moths.
Complete step by step solution:
To protect silk clothes and woollen clothes we use naphthalene balls.
When the clothes are kept for a long time the insects start to ruin the material of the clothes. So, these clothes have to be protected from insects. Therefore, we use naphthalene balls.
The naphthalene balls are insect repellant, so these balls can be directly kept between the clothes. The naphthalene has a property of insect repellent because of its odour.
Naphthalene is an aromatic hydrocarbon that has a formula ${{C}_{10}}{{H}_{8}}$. The structure of naphthalene is given below:

It has a concentrated odour which penetrates very easily. This odour makes it useful as an insect or moth repellent.
The naphthalene balls have the property for getting converted into gas directly from solid. This process is called sublimation. So what happens is the solid naphthalene balls kept in the clothes start to get directly converted into gas. This gas is a toxic gas, which is used to kill or repel insects. As these balls directly convert into gas, it leaves no residue.
So the correct answer is an option (D)- Naphthalene balls.
Note: Not only in clothes but also in washrooms, cupboards, etc. naphthalene balls are used for protection from insects. These naphthalene balls are toxic for both adult and larva form of moths.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
What is the median of the first 10 natural numbers class 10 maths CBSE

Which women's tennis player has 24 Grand Slam singles titles?

Who is the Brand Ambassador of Incredible India?

Why is there a time difference of about 5 hours between class 10 social science CBSE

Write a letter to the principal requesting him to grant class 10 english CBSE

A moving boat is observed from the top of a 150 m high class 10 maths CBSE




