
Which gives only glucose by hydrolysis?
A. Sucrose
B. Raffinose
C. Maltose
D. Galactose
Answer
588k+ views
Hint: In hydrolysis a molecule of water is added to the chemical compound. This causes both the compound and water to split up. In such a reaction the parent product gains a hydrogen ion. Hydrolysis reaction is the reverse of condensation reaction in which two compounds join together leaving behind a water molecule.
Complete step by step answer: In this question we have to find the compound which yields only glucose on hydrolysis. Hydrolysis is a reaction in which a reactant is made to split into two or more products with the addition of water molecules. In this reaction the parent product gets hydrogen ions.
Glucose is a type of sugar. Its chemical formula is ${C_6}{H_{12}}{O_6}$.
Sucrose is a disaccharide means it is composed of two monosaccharide. Monosaccharide is simple sugars. These are the simplest unit of carbohydrates. These can’t be hydrolyzed further. Chemical formula of sucrose is ${C_{12}}{H_{22}}{O_{11}}$. When hydrolyzed sucrose gives two products that is: glucose and fructose.
$\
{C_{12}}{H_{22}}{O_{11}} + {H_2}O\xrightarrow{{HCl}}{C_6}{H_{12}}{O_6} + {C_6}{H_{12}}{O_6} \\
{\text{Sucrose Glucose Fructose}} \\
\ $
Chemical formula of glucose and fructose is the same but the difference lies in their structure.
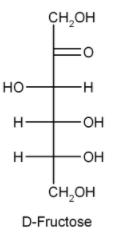
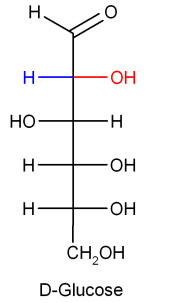
This is the simple difference in the structure of glucose and fructose. As we can see sucrose on hydrolysis gives two products but we need only one that is glucose. This means sucrose is not the answer.
Raffinose is a trisaccharide. Means it is made up of three monosaccharides. Chemical formula of raffinose is ${C_{18}}{H_{32}}{O_{16}}$ . Raffinose on hydrolysis yields galactose, glucose and fructose.
$\
{C_{18}}{H_{32}}{O_{16}} + {H_2}O\xrightarrow{{}}{C_6}{H_{12}}{O_6} + {C_6}{H_{12}}{O_6} + {C_6}{H_{12}}{O_6} \\
{\text{Raffinose Glucose Fructose Galactose}} \\
\ $
We need only glucose as the product but three products are coming from hydrolysis of Raffinose. This means raffinose is also not the answer.
Maltose is a disaccharide. Chemical formula of maltose is ${C_{12}}{H_{22}}{O_{11}}$ . On hydrolysis of maltose we get two molecules of glucose.
$\
{C_{12}}{H_{22}}{O_{11}} + {H_2}O\xrightarrow{{HCl}}2{C_6}{H_{12}}{O_6} \\
{\text{Maltose Glucose }} \\
\ $
So, this is our required product that is glucose. We needed a compound which gives only glucose as a product.
So, the correct answer is “Option C”.
Note: Monosaccharides are the simplest unit of carbohydrates. These can’t be hydrolyzed further. Disaccharides are made up of two monosaccharides. This means on hydrolysis of disaccharides we get two monosaccharides. Similarly there exist trisaccharides and polysaccharides as well.
Complete step by step answer: In this question we have to find the compound which yields only glucose on hydrolysis. Hydrolysis is a reaction in which a reactant is made to split into two or more products with the addition of water molecules. In this reaction the parent product gets hydrogen ions.
Glucose is a type of sugar. Its chemical formula is ${C_6}{H_{12}}{O_6}$.
Sucrose is a disaccharide means it is composed of two monosaccharide. Monosaccharide is simple sugars. These are the simplest unit of carbohydrates. These can’t be hydrolyzed further. Chemical formula of sucrose is ${C_{12}}{H_{22}}{O_{11}}$. When hydrolyzed sucrose gives two products that is: glucose and fructose.
$\
{C_{12}}{H_{22}}{O_{11}} + {H_2}O\xrightarrow{{HCl}}{C_6}{H_{12}}{O_6} + {C_6}{H_{12}}{O_6} \\
{\text{Sucrose Glucose Fructose}} \\
\ $
Chemical formula of glucose and fructose is the same but the difference lies in their structure.


This is the simple difference in the structure of glucose and fructose. As we can see sucrose on hydrolysis gives two products but we need only one that is glucose. This means sucrose is not the answer.
Raffinose is a trisaccharide. Means it is made up of three monosaccharides. Chemical formula of raffinose is ${C_{18}}{H_{32}}{O_{16}}$ . Raffinose on hydrolysis yields galactose, glucose and fructose.
$\
{C_{18}}{H_{32}}{O_{16}} + {H_2}O\xrightarrow{{}}{C_6}{H_{12}}{O_6} + {C_6}{H_{12}}{O_6} + {C_6}{H_{12}}{O_6} \\
{\text{Raffinose Glucose Fructose Galactose}} \\
\ $
We need only glucose as the product but three products are coming from hydrolysis of Raffinose. This means raffinose is also not the answer.
Maltose is a disaccharide. Chemical formula of maltose is ${C_{12}}{H_{22}}{O_{11}}$ . On hydrolysis of maltose we get two molecules of glucose.
$\
{C_{12}}{H_{22}}{O_{11}} + {H_2}O\xrightarrow{{HCl}}2{C_6}{H_{12}}{O_6} \\
{\text{Maltose Glucose }} \\
\ $
So, this is our required product that is glucose. We needed a compound which gives only glucose as a product.
So, the correct answer is “Option C”.
Note: Monosaccharides are the simplest unit of carbohydrates. These can’t be hydrolyzed further. Disaccharides are made up of two monosaccharides. This means on hydrolysis of disaccharides we get two monosaccharides. Similarly there exist trisaccharides and polysaccharides as well.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




