
Which of the following is a diatomic gas?
(A) Hydrogen
(B) Oxygen
(C) Chlorine
(D) All of the above
Answer
529.8k+ views
Hint: In a free state and at standard temperature and pressure, some elements exist as diatomic molecules to achieve stability. In these diatomic molecules, both the atoms are of the same element and hence are homonuclear.
Complete answer:
To determine whether the gas exists as a diatomic molecule, we need to check the stability of the diatomic molecule formed.
(A) Hydrogen atom has 1 electron in its valence shell and needs 1 more electron to form a complete outermost shell. So when two hydrogen atoms form a bond, both the hydrogen atoms share two electrons between them, forming a covalent single bond and completing the octet.
Its Lewis structure is as follows:

Hence ${{H}_{2}}$ is stable and exists as a diatomic gas.
(B) Oxygen atom has 6 electrons in its valence shell and needs 2 more electrons to form a complete outermost octet shell. So, when 2 oxygen atoms form a bond, both the oxygen atoms share 4 electrons between them, forming a covalent double bond and completing the octet.
Its Lewis structure is as follows:
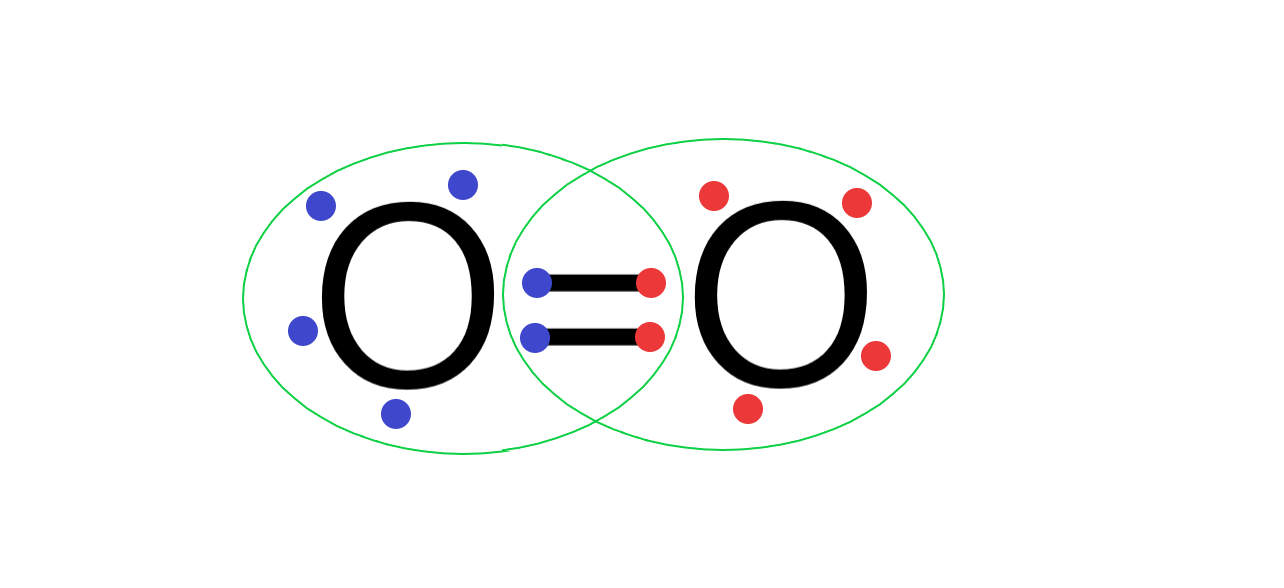
Hence ${{O}_{2}}$ is stable and exists as a diatomic gas.
(C) Chlorine atom has 7 electrons in its valence shell and needs 1 more electron to form a complete outermost octet shell. So when 2 chlorine atoms form a bond, both the chlorine atoms share 2 electrons between them, forming a covalent single bond and completing the octet.
Its Lewis structure is as follows:

Hence $C{{l}_{2}}$ is stable and exists as a diatomic gas.
So, the correct answer is option (D) All of the above.
Note:
It should be noted that other than chlorine $C{{l}_{2}}$, oxygen ${{O}_{2}}$, and hydrogen ${{H}_{2}}$, the only other gases that exist as homonuclear diatomic molecules at STP are fluorine ${{F}_{2}}$ and nitrogen ${{N}_{2}}$.
At temperatures higher than STP, bromine $B{{r}_{2}}$ , and iodine ${{I}_{2}}$ also exist as diatomic molecules.
Complete answer:
To determine whether the gas exists as a diatomic molecule, we need to check the stability of the diatomic molecule formed.
(A) Hydrogen atom has 1 electron in its valence shell and needs 1 more electron to form a complete outermost shell. So when two hydrogen atoms form a bond, both the hydrogen atoms share two electrons between them, forming a covalent single bond and completing the octet.
Its Lewis structure is as follows:

Hence ${{H}_{2}}$ is stable and exists as a diatomic gas.
(B) Oxygen atom has 6 electrons in its valence shell and needs 2 more electrons to form a complete outermost octet shell. So, when 2 oxygen atoms form a bond, both the oxygen atoms share 4 electrons between them, forming a covalent double bond and completing the octet.
Its Lewis structure is as follows:

Hence ${{O}_{2}}$ is stable and exists as a diatomic gas.
(C) Chlorine atom has 7 electrons in its valence shell and needs 1 more electron to form a complete outermost octet shell. So when 2 chlorine atoms form a bond, both the chlorine atoms share 2 electrons between them, forming a covalent single bond and completing the octet.
Its Lewis structure is as follows:

Hence $C{{l}_{2}}$ is stable and exists as a diatomic gas.
So, the correct answer is option (D) All of the above.
Note:
It should be noted that other than chlorine $C{{l}_{2}}$, oxygen ${{O}_{2}}$, and hydrogen ${{H}_{2}}$, the only other gases that exist as homonuclear diatomic molecules at STP are fluorine ${{F}_{2}}$ and nitrogen ${{N}_{2}}$.
At temperatures higher than STP, bromine $B{{r}_{2}}$ , and iodine ${{I}_{2}}$ also exist as diatomic molecules.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

What organs are located on the left side of your body class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




