
Which of the following is not the state of matter?
A.Solid
B.Liquid
C.Gas
D.Vapor
Answer
584.1k+ views
Hint: All the materials that we see in our daily life is made up of matter. There are basically three main states of matter and these states are classified on the basis of intermolecular forces and the arrangement of particles. Further, the states can be converted from one state to another by changing certain environmental factors.
Complete step by step answer:
The three primary states of matter are solid, liquid and gaseous states. These three forms of matter can be converted from one state of matter to another state by changing certain environmental factors such as temperature, pressure etc. One of the common examples is that ice can be converted from a solid into liquid water by increasing the temperature. Now let’s discuss each state in detail.
1.Solids- It is one of the fundamental states of matter. In this state the molecules are tightly packed because of the strong intermolecular forces. This state has a definite shape and volume and has a rigid structure. Example iron
2.Liquids-In this state, the molecules are closely packed due to weak intermolecular forces. These forces are weaker than solids but stronger than gases. In general, the conversion of solids into liquids takes place when we increase the temperature of solids to a point where they begin to melt. Example water
3.Gases- In this state, the distances between the molecules are large and the intermolecular forces are negligible. Moreover, they do not have a fixed shape or volume. Example ${O_2}$
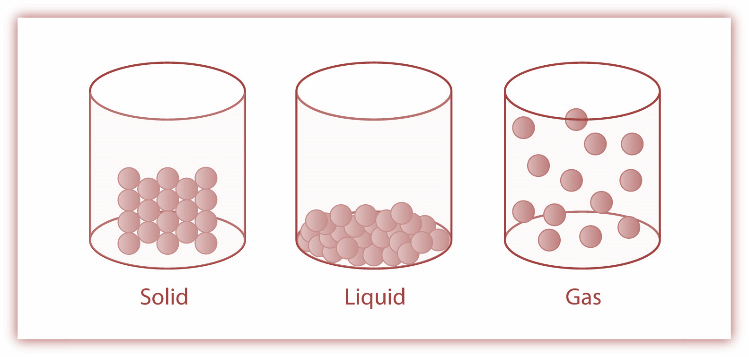
So, among the given options vapor is matter but not a state of matter. It is basically the type of substance which is formed when liquid converts into the gaseous phase.
Hence, option D is correct.
Note: Matter is composed of small particles known as atoms. These are so small and we can’t see them. These atoms come together and make the substances that we can see in our day to day life. Moreover, light is not a matter, it is basically a form of energy or we can say that it is electromagnetic radiation.
Complete step by step answer:
The three primary states of matter are solid, liquid and gaseous states. These three forms of matter can be converted from one state of matter to another state by changing certain environmental factors such as temperature, pressure etc. One of the common examples is that ice can be converted from a solid into liquid water by increasing the temperature. Now let’s discuss each state in detail.
1.Solids- It is one of the fundamental states of matter. In this state the molecules are tightly packed because of the strong intermolecular forces. This state has a definite shape and volume and has a rigid structure. Example iron
2.Liquids-In this state, the molecules are closely packed due to weak intermolecular forces. These forces are weaker than solids but stronger than gases. In general, the conversion of solids into liquids takes place when we increase the temperature of solids to a point where they begin to melt. Example water
3.Gases- In this state, the distances between the molecules are large and the intermolecular forces are negligible. Moreover, they do not have a fixed shape or volume. Example ${O_2}$

So, among the given options vapor is matter but not a state of matter. It is basically the type of substance which is formed when liquid converts into the gaseous phase.
Hence, option D is correct.
Note: Matter is composed of small particles known as atoms. These are so small and we can’t see them. These atoms come together and make the substances that we can see in our day to day life. Moreover, light is not a matter, it is basically a form of energy or we can say that it is electromagnetic radiation.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

10 examples of friction in our daily life




