
Which of the following shows \[ds{{p}^{2}}\] hybridisation and a square planar geometry?
A. $S{{F}_{6}}$
B. $Br{{F}_{5}}$
C. $PC{{l}_{5}}$
D. ${{\left[ Ni{{\left( CN \right)}_{4}} \right]}^{2-}}$
Answer
233.1k+ views
Hint: We have to find out the hybridisation, which means when different atomic orbitals are combined having different energies to give the equivalent orbitals. Here we will see the combination of one d orbital, one s orbital and two p orbitals to give the \[ds{{p}^{2}}\] hybridisation and square planar geometry.
Step by step solution:
- We will find the electronic configuration of Ni in the compound ${{\left[ Ni{{\left( CN \right)}_{4}} \right]}^{2-}}$,
- The electronic configuration of Ni is-$1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{8}}$
- And the electronic configuration of \[N{{i}^{2+}}\]will be-\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{0}}3{{d}^{8}}\]
- We can represent the valence bond representation as:
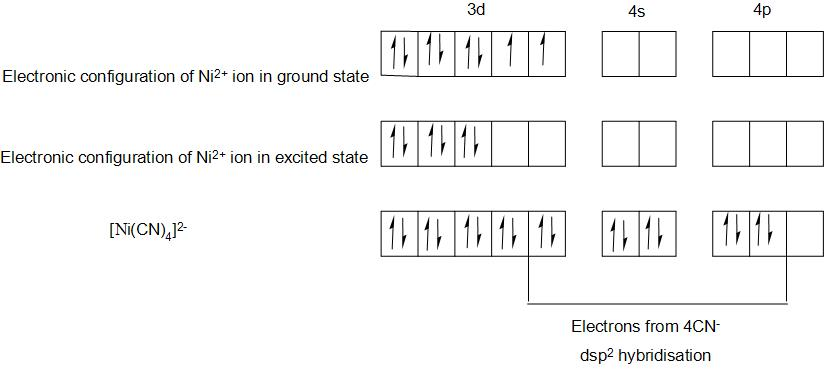
- Here we have filled the 8 electrons of \[N{{i}^{2+}}\] in 3d orbitals, the two unpaired electrons in the ground state of ion, will pair up in the excited state , due to the pairing energy supplied by the formation of strong bonds in the complex.
- This makes one of the 3d orbitals empty. By this we can see above that there are no unpaired electrons and hence the compound will be diamagnetic.
- Here we can see that 4 $C{{N}^{-}}$ ions will form a strong bond in the complex.
- And here the central metal ion undergoes \[ds{{p}^{2}}\] hybridisation and the complex ion takes square planar geometry.
- Therefore, we can conclude that the correct option is(d) that is ${{\left[ Ni{{\left( CN \right)}_{4}} \right]}^{2-}}$ shows \[ds{{p}^{2}}\] hybridisation and a square planar geometry.
- As we have seen from the above valence bond representation that there are no unpaired electrons present and hence it is diamagnetic, we can also say that it has zero magnetic moment.
Note:
- In presence of any strong field ligand like $C{{N}^{-}}$ , all the electrons are paired up, and in the presence of weak field ligands electrons are not paired up.
- We can calculate the magnetic moment, hybridisation, geometry, and magnetic nature from the valence bond representation.
Step by step solution:
- We will find the electronic configuration of Ni in the compound ${{\left[ Ni{{\left( CN \right)}_{4}} \right]}^{2-}}$,
- The electronic configuration of Ni is-$1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{8}}$
- And the electronic configuration of \[N{{i}^{2+}}\]will be-\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{0}}3{{d}^{8}}\]
- We can represent the valence bond representation as:

- Here we have filled the 8 electrons of \[N{{i}^{2+}}\] in 3d orbitals, the two unpaired electrons in the ground state of ion, will pair up in the excited state , due to the pairing energy supplied by the formation of strong bonds in the complex.
- This makes one of the 3d orbitals empty. By this we can see above that there are no unpaired electrons and hence the compound will be diamagnetic.
- Here we can see that 4 $C{{N}^{-}}$ ions will form a strong bond in the complex.
- And here the central metal ion undergoes \[ds{{p}^{2}}\] hybridisation and the complex ion takes square planar geometry.
- Therefore, we can conclude that the correct option is(d) that is ${{\left[ Ni{{\left( CN \right)}_{4}} \right]}^{2-}}$ shows \[ds{{p}^{2}}\] hybridisation and a square planar geometry.
- As we have seen from the above valence bond representation that there are no unpaired electrons present and hence it is diamagnetic, we can also say that it has zero magnetic moment.
Note:
- In presence of any strong field ligand like $C{{N}^{-}}$ , all the electrons are paired up, and in the presence of weak field ligands electrons are not paired up.
- We can calculate the magnetic moment, hybridisation, geometry, and magnetic nature from the valence bond representation.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

Hydrocarbons Class 11 Chemistry Chapter 9 CBSE Notes - 2025-26

Thermodynamics Class 11 Chemistry Chapter 5 CBSE Notes - 2025-26

Equilibrium Class 11 Chemistry Chapter 6 CBSE Notes - 2025-26

Organic Chemistry Some Basic Principles And Techniques Class 11 Chemistry Chapter 8 CBSE Notes - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 7 Redox Reactions (2025-26)




