
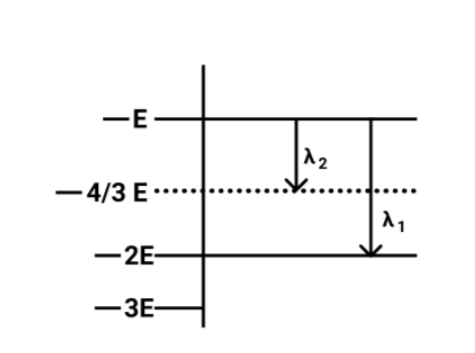
Some energy levels of a molecule are shown in fig. The ratio of the wavelengths $r = {\lambda _1}/{\lambda _2}$is given by:
$\left( a \right)$ $r = \dfrac{1}{3}$
$\left( b \right)$ $r = \dfrac{4}{3}$
$\left( c \right)$ $r = \dfrac{2}{3}$
$\left( d \right)$ $r = \dfrac{3}{4}$
Answer
233.1k+ views
Hint: we have been given two wavelengths and here are the two transitions. So assuming the corresponding two these two transitions, there is the emission of photons in each case. So firstly we will find the energy of that particular photon then using the relation which we have given in the formula, we will find the ratio between these two wavelengths.
Formula used
The energy of photons,
$E = \dfrac{{hc}}{\lambda }$
Here,
$E$, will be the energy
$h$, will be the planck's constant
$c$, will be the speed of light
$\lambda $, will be the wavelength
Complete Step By Step Solution: As we know the formula for the energy of the photon and it can be written as
$E = \dfrac{{hc}}{\lambda }$
And from here, $\lambda $can be written as
$ \Rightarrow \lambda = \dfrac{{hc}}{E}$
And hence we can say that
$ \Rightarrow \lambda \propto \dfrac{1}{E}$
Now from the figure,
The energy of a photon of wavelength ${\lambda _2}$will be equal to
$ \Rightarrow \vartriangle {E_2} = - E - \left( { - \dfrac{4}{3}} \right)$
On solving the above equation, we get
$ \Rightarrow \vartriangle {E_2} = \dfrac{E}{3}$
Now we will calculate the Energy of the photon of wavelength ${\lambda _1}$ and it will equal to
$ \Rightarrow \vartriangle {E_1} = - E - \left( { - 2E} \right)$
On solving the above equation, we get
$ \Rightarrow \vartriangle {E_1} = E$
So now we will calculate the ratios between the two energies
Therefore, it can be written as
$ \Rightarrow \dfrac{{{\lambda _1}}}{{{\lambda _2}}} = \dfrac{{\vartriangle {E_2}}}{{\vartriangle {E_1}}}$
Now on substituting the values, we get
$ \Rightarrow \dfrac{{{\lambda _1}}}{{{\lambda _2}}} = \dfrac{{\left( {\dfrac{E}{3}} \right)}}{E}$
So we will solve the final above equation to get the required ratios
$ \Rightarrow r = \dfrac{{{\lambda _1}}}{{{\lambda _2}}} = \dfrac{1}{3}$
Therefore, the option $a$will be the correct choice.
Note: According to Einstein's equation $E = m{c^2}$ anything that possesses energy has mass and anything that has mass has energy which is equal to the mass of the particle times square of the speed of light. Photons have $0$ rest mass but they have Energy which is equal to Planck constant times its frequency. So, if the photon has a fixed amount of energy which will be more than zero with zero rest mass but the photon does not exist which has $0$ moving mass so its energy will be zero.
Formula used
The energy of photons,
$E = \dfrac{{hc}}{\lambda }$
Here,
$E$, will be the energy
$h$, will be the planck's constant
$c$, will be the speed of light
$\lambda $, will be the wavelength
Complete Step By Step Solution: As we know the formula for the energy of the photon and it can be written as
$E = \dfrac{{hc}}{\lambda }$
And from here, $\lambda $can be written as
$ \Rightarrow \lambda = \dfrac{{hc}}{E}$
And hence we can say that
$ \Rightarrow \lambda \propto \dfrac{1}{E}$
Now from the figure,
The energy of a photon of wavelength ${\lambda _2}$will be equal to
$ \Rightarrow \vartriangle {E_2} = - E - \left( { - \dfrac{4}{3}} \right)$
On solving the above equation, we get
$ \Rightarrow \vartriangle {E_2} = \dfrac{E}{3}$
Now we will calculate the Energy of the photon of wavelength ${\lambda _1}$ and it will equal to
$ \Rightarrow \vartriangle {E_1} = - E - \left( { - 2E} \right)$
On solving the above equation, we get
$ \Rightarrow \vartriangle {E_1} = E$
So now we will calculate the ratios between the two energies
Therefore, it can be written as
$ \Rightarrow \dfrac{{{\lambda _1}}}{{{\lambda _2}}} = \dfrac{{\vartriangle {E_2}}}{{\vartriangle {E_1}}}$
Now on substituting the values, we get
$ \Rightarrow \dfrac{{{\lambda _1}}}{{{\lambda _2}}} = \dfrac{{\left( {\dfrac{E}{3}} \right)}}{E}$
So we will solve the final above equation to get the required ratios
$ \Rightarrow r = \dfrac{{{\lambda _1}}}{{{\lambda _2}}} = \dfrac{1}{3}$
Therefore, the option $a$will be the correct choice.
Note: According to Einstein's equation $E = m{c^2}$ anything that possesses energy has mass and anything that has mass has energy which is equal to the mass of the particle times square of the speed of light. Photons have $0$ rest mass but they have Energy which is equal to Planck constant times its frequency. So, if the photon has a fixed amount of energy which will be more than zero with zero rest mass but the photon does not exist which has $0$ moving mass so its energy will be zero.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding Uniform Acceleration in Physics

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

Dual Nature of Radiation and Matter Class 12 Physics Chapter 11 CBSE Notes - 2025-26

Understanding the Electric Field of a Uniformly Charged Ring

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Electromagnetic Waves and Their Importance




