
(a) Compound (A) with molecular formula ${C_2}{H_4}O$ reduces Tollen’s reagent. (A) on treatment with \[HCN\] gives compound (B). Compound(B) on hydrolysis with an acid gives compound (C) with molecular formula \[{C_3}{H_6}{O_3}\] which is an optically active compound. Compound (A) on reduction with \[{N_2}{H_4}/{C_2}{H_5}ONa\;\] gives a hydrocarbon (D) of molecular formula \[{C_2}{H_6}\]. Identify (A), (B), (C) and (D) and explain the reactions.
(b) Ionic conductance at infinite dilution of \[A{l^{^{3 + }}}\] and \[S0_4^{2 - }\] are \[189oh{m^{ - 1}}c{m^2}gmequi{v^{ - 1}}\] and \[160oh{m^{ - 1}}c{m^2}gmequi{v^{ - 1}}\].Calculate equivalent and molar conductance of electrolyte at infinite dilution.
Answer
569.7k+ views
Hint: -The reaction with Tollen’s reagent is given by aldehydes and alpha-hydroxy ketones only. The reaction with HCN gives cyanohydrins which hydrolyse in the presence of an acid to give carboxylic acids and if any carboxylic acid has a chiral carbon, it is an optically active compound.
-According to Kohlrausch’s law, the sum of conductances of all ions is equal to the conductance of an electrolyte. Molecular formula gives the number of ions that will be formed.
Complete Step by step answer:
Since the Tollen’s reagent (Ammoniacal silver ) is reduced by aldehydes or alpha-hydroxy ketones, the compound (A), having the formula \[{C_2}{H_4}O\] can only be an aldehyde and not alpha-hydroxy ketone as it contains only one oxygen atom. So, the structural formula of compound (A) will be that of an acetaldehyde or ethanal.
Compound (A) is acetaldehyde or \[C{H_3}CHO\]
The reaction of acetaldehyde with HCN will be a nucleophilic addition reaction which will give cyanohydrin. It is depicted below:
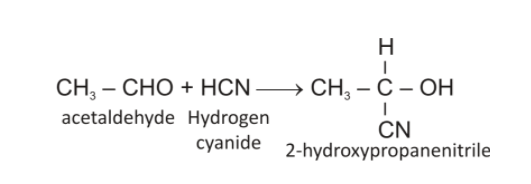
So, compound (B) is 2-hydroxypropanenitrile
Hydrolysis of nitriles gives carboxylic acid in which the\[CN\] group is changed to \[COOH\] group. Therefore, Compound (C) would be 2-hydroxypropanoic acid or commonly known as lactic acid.
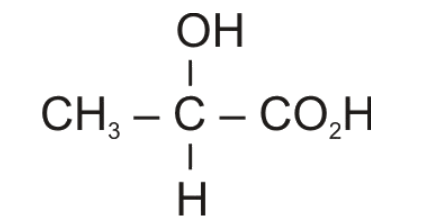
It is an optically active compound because alpha-carbon or the carbon directly attached to -\[COOH\] group is chiral carbon as it is attached to four different groups.
Compound (D) which is got by reduction of compound (A) by a strong reducing agent, \[{N_2}{H_4}\] or hydrazine, is ethane having the formula \[{C_2}{H_6}\]
Therefore, compound (A) is acetaldehyde, Compound (B) is 2-hydroxypropanenitrile, compound (C) is lactic acid, and compound (D) is ethane.
Given:
Ionic conductance of \[A{l^{^{3 + }}}\] at infinite dilution:
\[\lambda _{eq}^0\left( {A{l^{3 + }}} \right) = 189\;oh{m^{ - 1}}c{m^2}{\left( {g - equiv} \right)^{ - 1}}\]
Ionic conductance of \[S0_4^{2 - }\] at infinite dilution:
\[\lambda _{eq}^0\left( {SO_4^{2 - }} \right) = 160\;oh{m^{ - 1}}c{m^2}{\left( {g - equiv} \right)^{ - 1}}\]
Finding the equivalent conductance at infinite dilution
Now, the electrolyte in question is \[A{l_2}{\left( {S{O_4}} \right)_3}\]which contains two \[A{l^{^{3 + }}}\] ions and three \[S0_4^{2 - }\]ions
Therefore,
\[{{\Lambda }}_{eq}^0(A{l_2}{\left( {S{O_4}} \right)_3} = 2 \times \lambda _{eq}^0\left( {A{l^{3 + }}} \right) + 3 \times \lambda _{eq}^0\left( {SO_4^{2 - }} \right)\]
\[ = > {{\Lambda }}_{eq}^0\left( {A{l_2}{{\left( {S{O_4}} \right)}_3}} \right) = 2 \times 189 + 3 \times 160\]\[ = 858\;oh{m^{ - 1}}c{m^2}{\left( {g - equiv} \right)^{ - 1}}\]
This is the equivalent conductance of an electrolyte at infinite dilution.
Converting the equivalent conductance obtained in step 1 to molar conductance at infinite dilution
\[{{\Lambda }}_m^0 = 858\;\dfrac{{c{m^2}}}{{ohm}} \times \dfrac{1}{{\left( {g - equiv} \right)}} \times \dfrac{{6\;\left( {g - equiv} \right)}}{{1\;mol}} = 5148oh{m^{ - 1}}c{m^2}mo{l^{ - 1}}\]
1 mole of \[A{l_2}{\left( {S{O_4}} \right)_3}\]is equal to 6 g-equivalents as the total positive or negative valency is 6 in this particular electrolyte.
Note: - Remember that Tollen’s reagent is a weak oxidizing agent and it is reduced by only those carbonyl compounds in carbonyl group are not surrounded by alkyl groups, i.e. aldehydes or alpha-hydroxy ketones. Further, \[HCN\] will undergo breakage to give \[C{N^{ - 1}}\] which is a good nucleophile and will attack carbonyl carbon of aldehyde and since that carbon is unsaturated, it will be addition reaction. Further, hydrazine will reduce aldehyde into its corresponding alkane and not alcohol as it is a very strong reducing agent.
-The number of ions must be taken into account while adding the ionic conductances to get the overall electrolyte conductance. For this, the formula of an electrolyte must be deduced through valency exchange or criss-cross method. Molar conductance can be derived from equivalent conductance by just changing the units, i.e. (g-equiv) to mol. The knowledge that molar mass needs to be divided by total positive or total negative valency to get equivalent mass in case of ionic salts helps in conversion.
-According to Kohlrausch’s law, the sum of conductances of all ions is equal to the conductance of an electrolyte. Molecular formula gives the number of ions that will be formed.
Complete Step by step answer:
Since the Tollen’s reagent (Ammoniacal silver ) is reduced by aldehydes or alpha-hydroxy ketones, the compound (A), having the formula \[{C_2}{H_4}O\] can only be an aldehyde and not alpha-hydroxy ketone as it contains only one oxygen atom. So, the structural formula of compound (A) will be that of an acetaldehyde or ethanal.
Compound (A) is acetaldehyde or \[C{H_3}CHO\]
The reaction of acetaldehyde with HCN will be a nucleophilic addition reaction which will give cyanohydrin. It is depicted below:

So, compound (B) is 2-hydroxypropanenitrile
Hydrolysis of nitriles gives carboxylic acid in which the\[CN\] group is changed to \[COOH\] group. Therefore, Compound (C) would be 2-hydroxypropanoic acid or commonly known as lactic acid.

It is an optically active compound because alpha-carbon or the carbon directly attached to -\[COOH\] group is chiral carbon as it is attached to four different groups.
Compound (D) which is got by reduction of compound (A) by a strong reducing agent, \[{N_2}{H_4}\] or hydrazine, is ethane having the formula \[{C_2}{H_6}\]
Therefore, compound (A) is acetaldehyde, Compound (B) is 2-hydroxypropanenitrile, compound (C) is lactic acid, and compound (D) is ethane.
Given:
Ionic conductance of \[A{l^{^{3 + }}}\] at infinite dilution:
\[\lambda _{eq}^0\left( {A{l^{3 + }}} \right) = 189\;oh{m^{ - 1}}c{m^2}{\left( {g - equiv} \right)^{ - 1}}\]
Ionic conductance of \[S0_4^{2 - }\] at infinite dilution:
\[\lambda _{eq}^0\left( {SO_4^{2 - }} \right) = 160\;oh{m^{ - 1}}c{m^2}{\left( {g - equiv} \right)^{ - 1}}\]
Finding the equivalent conductance at infinite dilution
Now, the electrolyte in question is \[A{l_2}{\left( {S{O_4}} \right)_3}\]which contains two \[A{l^{^{3 + }}}\] ions and three \[S0_4^{2 - }\]ions
Therefore,
\[{{\Lambda }}_{eq}^0(A{l_2}{\left( {S{O_4}} \right)_3} = 2 \times \lambda _{eq}^0\left( {A{l^{3 + }}} \right) + 3 \times \lambda _{eq}^0\left( {SO_4^{2 - }} \right)\]
\[ = > {{\Lambda }}_{eq}^0\left( {A{l_2}{{\left( {S{O_4}} \right)}_3}} \right) = 2 \times 189 + 3 \times 160\]\[ = 858\;oh{m^{ - 1}}c{m^2}{\left( {g - equiv} \right)^{ - 1}}\]
This is the equivalent conductance of an electrolyte at infinite dilution.
Converting the equivalent conductance obtained in step 1 to molar conductance at infinite dilution
\[{{\Lambda }}_m^0 = 858\;\dfrac{{c{m^2}}}{{ohm}} \times \dfrac{1}{{\left( {g - equiv} \right)}} \times \dfrac{{6\;\left( {g - equiv} \right)}}{{1\;mol}} = 5148oh{m^{ - 1}}c{m^2}mo{l^{ - 1}}\]
1 mole of \[A{l_2}{\left( {S{O_4}} \right)_3}\]is equal to 6 g-equivalents as the total positive or negative valency is 6 in this particular electrolyte.
Note: - Remember that Tollen’s reagent is a weak oxidizing agent and it is reduced by only those carbonyl compounds in carbonyl group are not surrounded by alkyl groups, i.e. aldehydes or alpha-hydroxy ketones. Further, \[HCN\] will undergo breakage to give \[C{N^{ - 1}}\] which is a good nucleophile and will attack carbonyl carbon of aldehyde and since that carbon is unsaturated, it will be addition reaction. Further, hydrazine will reduce aldehyde into its corresponding alkane and not alcohol as it is a very strong reducing agent.
-The number of ions must be taken into account while adding the ionic conductances to get the overall electrolyte conductance. For this, the formula of an electrolyte must be deduced through valency exchange or criss-cross method. Molar conductance can be derived from equivalent conductance by just changing the units, i.e. (g-equiv) to mol. The knowledge that molar mass needs to be divided by total positive or total negative valency to get equivalent mass in case of ionic salts helps in conversion.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




