
Conversion of water to steam is accompanied by which process?
A. Adiabatic
B. Isothermal
C. Isochoric
D. Cyclic
Answer
609.3k+ views
Hint: First of all, we need to see what happens when water is converted into steam. We know that when a liquid state is changing into a gaseous state the temperature remains constant. And a constant temperature process is known as an isothermal process.
Complete step by step answer:
First, we will see what happens when water is converted to steam; that is a liquid state is converted to a gaseous state. When heat is continuously provided to the water its temperature starts to rise with time, up to a certain temperature. After that temperature when further heat is provided the heat energy is used up for breaking the intermolecular bonds in the liquid state and not for increasing the kinetic energy. Therefore the temperature remains constant whenever a change in phase occurs either from liquid to gas that is the boiling Point or from solid to a liquid that is the melting point as shown in figure 1.
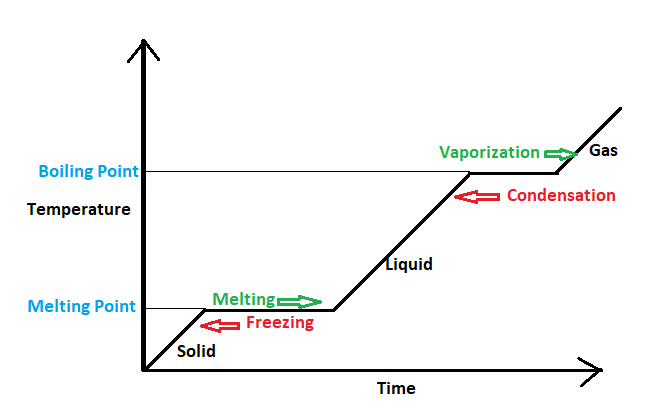
These types of Thermodynamic processes which take place with constant temperature are known as Isothermal processes. So conversion of water to steam is an Isothermal process hence option B is correct.
We will also see the definition of the other terms in the question as it will be helpful in future
A. An adiabatic process takes place without transferring heat or mass between a thermodynamic system and its surroundings.
C. An isochoric process is a thermodynamic process in which the volume of the enclosed system undergoing such a process remains unchanged.
D. A cyclic process is a thermodynamic process where the starting and ending state is the same.
Note: For these types of questions, we first need to recall the different thermodynamic processes and then see in which category the given question falls, that is we need to know how the processes are taking place and what are the parameters that are getting affected and what parameters remain constant.
Complete step by step answer:
First, we will see what happens when water is converted to steam; that is a liquid state is converted to a gaseous state. When heat is continuously provided to the water its temperature starts to rise with time, up to a certain temperature. After that temperature when further heat is provided the heat energy is used up for breaking the intermolecular bonds in the liquid state and not for increasing the kinetic energy. Therefore the temperature remains constant whenever a change in phase occurs either from liquid to gas that is the boiling Point or from solid to a liquid that is the melting point as shown in figure 1.

Figure 1
These types of Thermodynamic processes which take place with constant temperature are known as Isothermal processes. So conversion of water to steam is an Isothermal process hence option B is correct.
We will also see the definition of the other terms in the question as it will be helpful in future
A. An adiabatic process takes place without transferring heat or mass between a thermodynamic system and its surroundings.
C. An isochoric process is a thermodynamic process in which the volume of the enclosed system undergoing such a process remains unchanged.
D. A cyclic process is a thermodynamic process where the starting and ending state is the same.
Note: For these types of questions, we first need to recall the different thermodynamic processes and then see in which category the given question falls, that is we need to know how the processes are taking place and what are the parameters that are getting affected and what parameters remain constant.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
What is the median of the first 10 natural numbers class 10 maths CBSE

Which women's tennis player has 24 Grand Slam singles titles?

Who is the Brand Ambassador of Incredible India?

Why is there a time difference of about 5 hours between class 10 social science CBSE

Write a letter to the principal requesting him to grant class 10 english CBSE

State and prove converse of BPT Basic Proportionality class 10 maths CBSE




