
How would you convert ethyl bromide to butane?
Answer
552k+ views
Hint:We know that a reaction which has alkyl halide react with metallic sodium metal in the presence of a dry ether with solvent in order to form a symmetrical alkane which contains double number of a carbon atom present in alkyl halide as well as a name of reaction which is known as a wurtz reaction. It's used for preparation of higher alkane.
Complete step-by-step answer:Now, here we have to convert ethyl chloride to butane:
Here ethyl chloride react with sodium metal which in turn form a higher alkane and it is given by:
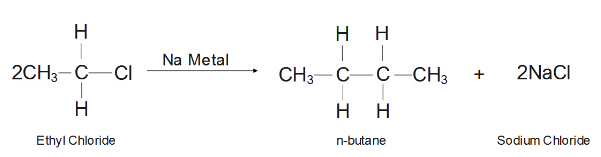
From above reaction two mole of ethyl chloride reacts with a sodium metal in order to form one mole of butane along with two mole of a sodium chloride as byproduct. This sort of reaction is known as wurtz reaction. The reaction occurs in presence of sodium metal wurtz reaction which is one of best methods to prepare a higher alkane from the alkyl halide.
Thus, Wurtz reaction generally fails with a tertiary alkyl halide since under basic conditions of reaction so they prefer to undergo a dehydrohalogenation in order to form alkene. When the mixture of two different alkyl halides is used all three possible alkane are formed.
Note: Note that alkyl halide which have lot of application in an organic chemistry as well as by using alkyl halide so we can prepare alkane which they have same number of a carbon atom side by side we could prepare higher alkane by using a sodium metal through wurtz reaction.
Complete step-by-step answer:Now, here we have to convert ethyl chloride to butane:
Here ethyl chloride react with sodium metal which in turn form a higher alkane and it is given by:

From above reaction two mole of ethyl chloride reacts with a sodium metal in order to form one mole of butane along with two mole of a sodium chloride as byproduct. This sort of reaction is known as wurtz reaction. The reaction occurs in presence of sodium metal wurtz reaction which is one of best methods to prepare a higher alkane from the alkyl halide.
Thus, Wurtz reaction generally fails with a tertiary alkyl halide since under basic conditions of reaction so they prefer to undergo a dehydrohalogenation in order to form alkene. When the mixture of two different alkyl halides is used all three possible alkane are formed.
Note: Note that alkyl halide which have lot of application in an organic chemistry as well as by using alkyl halide so we can prepare alkane which they have same number of a carbon atom side by side we could prepare higher alkane by using a sodium metal through wurtz reaction.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




