
Coupling of benzene diazonium chloride with 1-naphthol in alkaline medium will give:
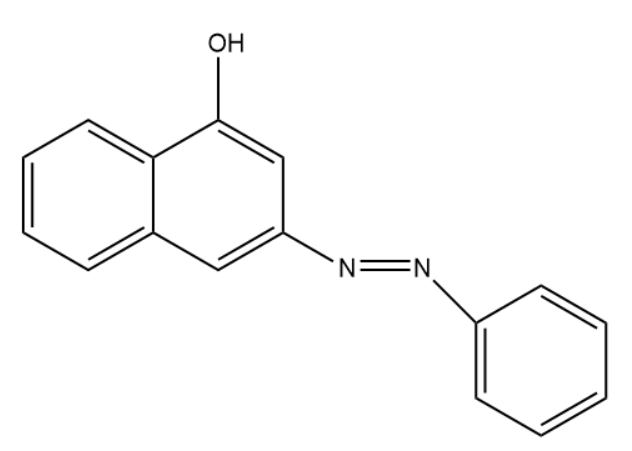
(A)
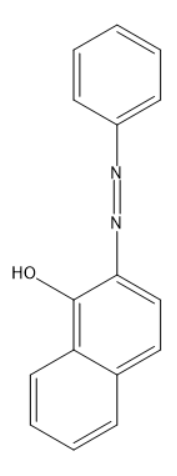
(B)
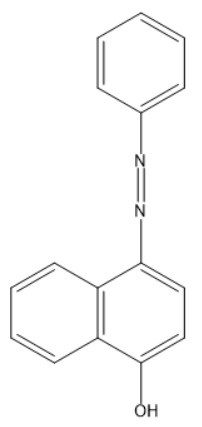
C)
D)
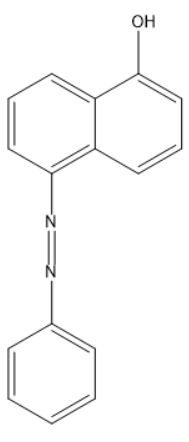




Answer
570k+ views
Hint: It is a type of azo coupling. In such reactions diazonium salt shows electrophilic attack on other aromatic compounds and substitution reaction occurs.
Complete Solution :
Azo coupling: Those reactions in which a diazonium compound reacts with another aromatic compound. The product of such reactions is an azo compound. These reactions come under the category of electrophilic aromatic substitution reaction, in which aryl diazonium cation behaves as electrophile and attack on activated arene which is a nucleophile.
- The given question is asking about the product of reaction between benzene diazonium chloride and 1-naphthol (alpha naphthol). So, the Product is this reaction is a dye organol brown. In this dye the electrophile aryl diazonium cation substitutes the hydrogen of at para position to the hydroxyl group of alpha naphthol among the three open positions i.e. ortho, meta, and para to the hydroxyl group.
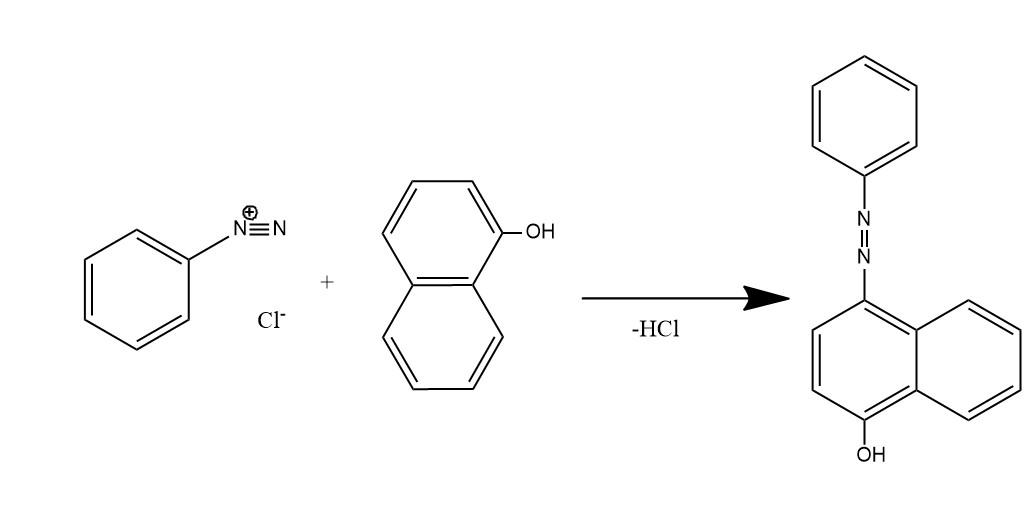
Substitution occurs at para position of naphthol ring because of two reasons:
(1) Hydroxyl groups of phenols are ortho and para directing for electrophilic substitution reactions.
(2) In naphthalene system electrophilic substitution is kinetically favored next to the other ring.
So, among ortho and para positions, para one is next to the ring which favors the substitution at para position.
So, the correct answer is “Option C”.
Note: Organol brown is also called hex type brown, a Chemical name is 4-(Phenylazo)-1-naphthol, Chemical formula is ${{C}_{16}}{{H}_{12}}{{N}_{2}}O$.
Complete Solution :
Azo coupling: Those reactions in which a diazonium compound reacts with another aromatic compound. The product of such reactions is an azo compound. These reactions come under the category of electrophilic aromatic substitution reaction, in which aryl diazonium cation behaves as electrophile and attack on activated arene which is a nucleophile.
- The given question is asking about the product of reaction between benzene diazonium chloride and 1-naphthol (alpha naphthol). So, the Product is this reaction is a dye organol brown. In this dye the electrophile aryl diazonium cation substitutes the hydrogen of at para position to the hydroxyl group of alpha naphthol among the three open positions i.e. ortho, meta, and para to the hydroxyl group.

Substitution occurs at para position of naphthol ring because of two reasons:
(1) Hydroxyl groups of phenols are ortho and para directing for electrophilic substitution reactions.
(2) In naphthalene system electrophilic substitution is kinetically favored next to the other ring.
So, among ortho and para positions, para one is next to the ring which favors the substitution at para position.

So, the correct answer is “Option C”.
Note: Organol brown is also called hex type brown, a Chemical name is 4-(Phenylazo)-1-naphthol, Chemical formula is ${{C}_{16}}{{H}_{12}}{{N}_{2}}O$.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




