
EDTA is a multidentate ligand. Its denticity or multiplicity is ………………………
Answer
539.4k+ views
Hint :Ligands are the donor atoms, molecules or anions which donate a pair of electrons to metal atoms or ions are called ligands. In EDTA there are four Oxygen atoms and two Nitrogen atoms. Nitrogen and oxygen both have lone pairs to coordinate with the central atom in the complex compound.
Complete Step By Step Answer:
We know that Denticity refers to the number of donor groups in a particular ligand that transfer lone pairs to the central atom and bind it in complex compounds.
In most of the cases, only a single atom in the ligand binds to the metal, then the denticity equals one, and the ligand is said to be monodentate or bidentate.
Ethylene diamine tetra acetate ions (EDTA) form a complex with metal ions in coordination compounds. Denticity refers to the number of donor groups that are attached to the ligand to participate in forming coordinate bonds with the complex compounds.
EDTA is a type of ligand in which donor atoms contain lone pairs of electrons and can donate electrons to positively charged central atom species to form complex compounds. EDTA is a multidentate ligand. Its denticity (multiplicity) is 6.
In EDTA, there are four Oxygen atoms and two Nitrogen atoms. Nitrogen and oxygen both have lone pairs to coordinate with the central atom in the complex compound. EDTA is also used extensively in the analysis of blood. It is an anticoagulant for blood samples in the field of medicine.
Structure of EDTA:
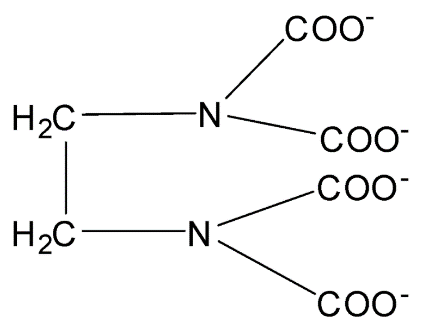
EDTA is a polydentate ligand have flexidentate character in which four Oxygen atoms and two Nitrogen atoms form coordinate bonds with the central metal atom or ion. Therefore, there are six donor atoms present in EDTA.
Hence, EDTA is a hexadentate ligand. Its denticity or multiplicity is 6.
Note :
The coordination compounds are of great importance and are widely present in minerals, plants and animals and are known to play many important functions in analytical chemistry, metallurgy, biological systems, industry and medicine. EDTA is used in treatment of lead poisoning. EDTA which is a hexadentate ligand also acts as tetradentate or pentadentate in certain complexes.
Complete Step By Step Answer:
We know that Denticity refers to the number of donor groups in a particular ligand that transfer lone pairs to the central atom and bind it in complex compounds.
In most of the cases, only a single atom in the ligand binds to the metal, then the denticity equals one, and the ligand is said to be monodentate or bidentate.
Ethylene diamine tetra acetate ions (EDTA) form a complex with metal ions in coordination compounds. Denticity refers to the number of donor groups that are attached to the ligand to participate in forming coordinate bonds with the complex compounds.
EDTA is a type of ligand in which donor atoms contain lone pairs of electrons and can donate electrons to positively charged central atom species to form complex compounds. EDTA is a multidentate ligand. Its denticity (multiplicity) is 6.
In EDTA, there are four Oxygen atoms and two Nitrogen atoms. Nitrogen and oxygen both have lone pairs to coordinate with the central atom in the complex compound. EDTA is also used extensively in the analysis of blood. It is an anticoagulant for blood samples in the field of medicine.
Structure of EDTA:

EDTA is a polydentate ligand have flexidentate character in which four Oxygen atoms and two Nitrogen atoms form coordinate bonds with the central metal atom or ion. Therefore, there are six donor atoms present in EDTA.
Hence, EDTA is a hexadentate ligand. Its denticity or multiplicity is 6.
Note :
The coordination compounds are of great importance and are widely present in minerals, plants and animals and are known to play many important functions in analytical chemistry, metallurgy, biological systems, industry and medicine. EDTA is used in treatment of lead poisoning. EDTA which is a hexadentate ligand also acts as tetradentate or pentadentate in certain complexes.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




