
Explain the Haber's process of manufacturing Ammonia with a neat diagram.
Answer
579.6k+ views
Hint: The Haber process also known as Haber–Bosch process is a process of an artificial nitrogen fixation and is considered to be the main industrial phenomenon for ammonia production. The process is named after its German inventors namely Carl Bosch and Fritz Haber.
Complete step by step answer:
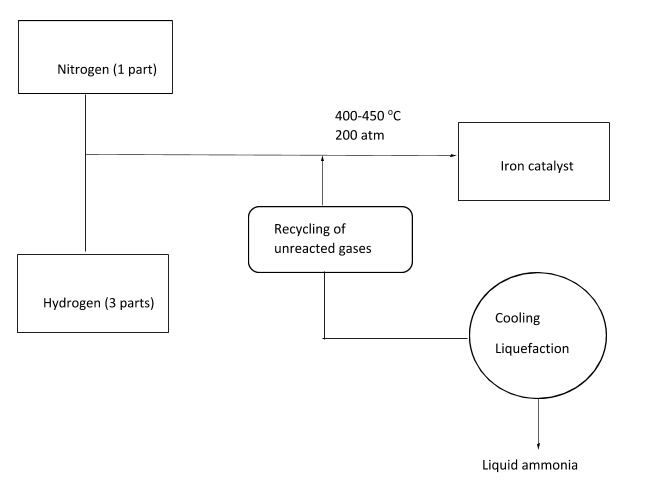
The Haber’s process combines nitrogen with hydrogen in the ratio of 1:3 in order to produce ammonia. Nitrogen is supplied from air while the major source for hydrogen is methane from the natural gas. Reversible reaction takes place and the generation of ammonia is an exothermic reaction. This reaction taking place in the reactor at temperature of 400-450 oC and 200 atm pressure can be written in the following manner:
\[{N_2}\left( g \right){\text{ }} + {\text{ }}{H_2}\left( g \right){\text{ }} \rightleftharpoons {\text{ }}2N{H_3}\left( g \right)\]
In this reaction, iron is generally utilized as a catalyst. At each stage of the passage of gases through a reactor, only around 15% of nitrogen as well as hydrogen are converted to ammonia. By continuously recycling the unreacted nitrogen as well as hydrogen, 98% of ammonia can be obtained. In the reaction, nitrogen can be attained by the process of liquefaction by removing nitrogen from soil. On the other hand, hydrogen can be attained by the process of steam reforming from the natural gas.
$C{H_4}(g) + {H_2}O \to {H_2}(g) + CO(g)$
The Haber process can be better understood with the following diagram:
Note:
The generation of ammonia through the Haber process is the most energy-intensive commodity chemical which is responsible for almost 1%–2% of consumption of global energy and 1.44% of \[C{O_2}\] emissions.
Complete step by step answer:
The Haber’s process combines nitrogen with hydrogen in the ratio of 1:3 in order to produce ammonia. Nitrogen is supplied from air while the major source for hydrogen is methane from the natural gas. Reversible reaction takes place and the generation of ammonia is an exothermic reaction. This reaction taking place in the reactor at temperature of 400-450 oC and 200 atm pressure can be written in the following manner:
\[{N_2}\left( g \right){\text{ }} + {\text{ }}{H_2}\left( g \right){\text{ }} \rightleftharpoons {\text{ }}2N{H_3}\left( g \right)\]
In this reaction, iron is generally utilized as a catalyst. At each stage of the passage of gases through a reactor, only around 15% of nitrogen as well as hydrogen are converted to ammonia. By continuously recycling the unreacted nitrogen as well as hydrogen, 98% of ammonia can be obtained. In the reaction, nitrogen can be attained by the process of liquefaction by removing nitrogen from soil. On the other hand, hydrogen can be attained by the process of steam reforming from the natural gas.
$C{H_4}(g) + {H_2}O \to {H_2}(g) + CO(g)$
The Haber process can be better understood with the following diagram:

Note:
The generation of ammonia through the Haber process is the most energy-intensive commodity chemical which is responsible for almost 1%–2% of consumption of global energy and 1.44% of \[C{O_2}\] emissions.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




