
What happens when
(i) Chlorobenzene is treated with \[C{l_2}/FeC{l_3}\]
(ii) Ethyl chloride is treated with \[AgN{O_2}\]
(iii) 2-bromopentane is treated with alcoholic KOH?
Write the chemical equation in support of your answer.
Answer
558.3k+ views
Hint: In the first reaction, when chlorobenzene reacts with \[C{l_2}/FeC{l_3}\], it will undergo nucleophilic substitution reaction and give multiple products.
In the second reaction, Ethyl chloride is reacted with \[AgN{O_2}\]. This reaction is the Victor Mayer method.
In the third reaction, 2-bromopentane is treated with alcoholic KOH, will follow Saytzeff’s rule and undergo \[\beta - \] elimination.
Complete step by step answer:
(i) When Chlorobenzene is treated with \[C{l_2}/FeC{l_3}\], it will lead to the formation of both ortho and para isomers. The ortho isomer will be 1,2-Dichlorobenzene and the para isomer will be 1,4-Dichlorobenzene. The reaction is given below
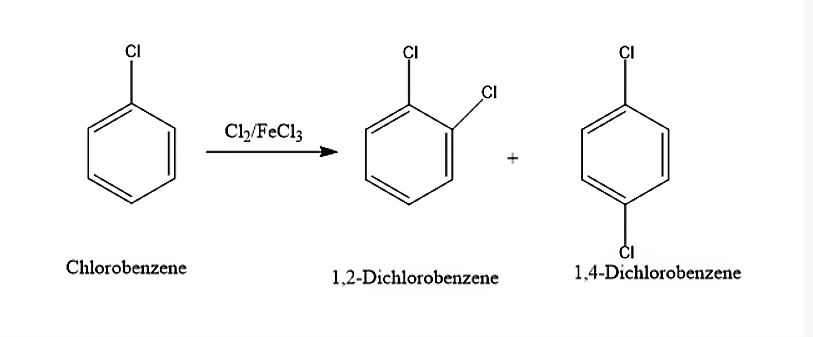
(ii) When Ethyl chloride, \[C{H_3}C{H_2}Cl\] is treated with Silver Nitrite, \[AgN{O_2}\]it will lead to the formation of Nitroethane and white precipitate of Silver chloride, \[AgCl\]. This process is known as Victor Meyer Method.
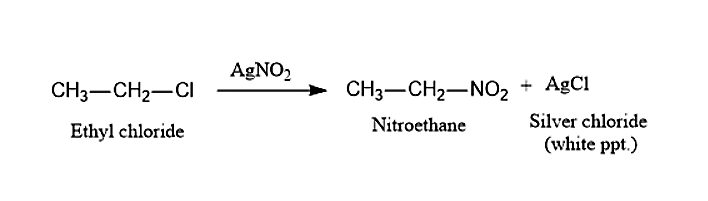
(iii) When 2-bromopentane is treated with alcoholic KOH it will follow Saytzeff’s rule and will undergo \[\beta - \] elimination. This reaction will give two products. Among that one will be major and the other will be minor. The major product in this reaction is Pent-2-ene and the minor product will be Pent-1-ene.

Therefore,
(i) When Chlorobenzene is treated with \[C{l_2}/FeC{l_3}\], it will give 1,2-Dichlorobenzene and 1,4-Dichlorobenzene.
(ii) When Ethyl chloride is treated with \[AgN{O_2}\], it will give Nitroethane.
(iii) When 2-bromopentane is treated with alcoholic KOH, it will give Pent-2-ene as the major product and Pent-1-ene as the minor product.
Note: According to the Saytzeff’s rule, when dehydrohalogenation reaction takes place and the most stable product will be that alkene i.e., 2-pentene, which has largely substituted alkene group in it. The less substituted alkene will be the minor product and also called the Hofmann product.
In the second reaction, Ethyl chloride is reacted with \[AgN{O_2}\]. This reaction is the Victor Mayer method.
In the third reaction, 2-bromopentane is treated with alcoholic KOH, will follow Saytzeff’s rule and undergo \[\beta - \] elimination.
Complete step by step answer:
(i) When Chlorobenzene is treated with \[C{l_2}/FeC{l_3}\], it will lead to the formation of both ortho and para isomers. The ortho isomer will be 1,2-Dichlorobenzene and the para isomer will be 1,4-Dichlorobenzene. The reaction is given below

(ii) When Ethyl chloride, \[C{H_3}C{H_2}Cl\] is treated with Silver Nitrite, \[AgN{O_2}\]it will lead to the formation of Nitroethane and white precipitate of Silver chloride, \[AgCl\]. This process is known as Victor Meyer Method.

(iii) When 2-bromopentane is treated with alcoholic KOH it will follow Saytzeff’s rule and will undergo \[\beta - \] elimination. This reaction will give two products. Among that one will be major and the other will be minor. The major product in this reaction is Pent-2-ene and the minor product will be Pent-1-ene.

Therefore,
(i) When Chlorobenzene is treated with \[C{l_2}/FeC{l_3}\], it will give 1,2-Dichlorobenzene and 1,4-Dichlorobenzene.
(ii) When Ethyl chloride is treated with \[AgN{O_2}\], it will give Nitroethane.
(iii) When 2-bromopentane is treated with alcoholic KOH, it will give Pent-2-ene as the major product and Pent-1-ene as the minor product.
Note: According to the Saytzeff’s rule, when dehydrohalogenation reaction takes place and the most stable product will be that alkene i.e., 2-pentene, which has largely substituted alkene group in it. The less substituted alkene will be the minor product and also called the Hofmann product.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




