
Name the experiment illustrated aside.
A.Fountain experiment
B.Lavoisier experiment
C.Blaise Pascal carries
D.None of these

Answer
571.2k+ views
Hint: The main components here are hydrogen chloride, blue litmus . The experiment process has a characteristic effect which gave the experiment its name. This effect has a striking resemblance to a geological feature.
Complete answer:
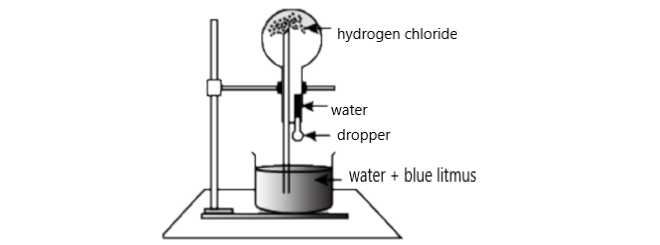
At first we have to look into the fountain experiment.
This method is done by introducing water through an inlet to a container with hydrogen chloride gas.
Then the gas gets mixed with water and we can see gradually the pressure inside the container drops down.
Then more water is forced to enter the container from another inlet producing a fountain effect.
This experiment is used to introduce concepts like solubility and gas laws.
Then blue litmus is added to check the acidic nature of hydrogen chloride gas in the round bottom flask.
From the definition of this experiment, it clearly defines the highlighted components like hydrogen chloride, blue litmus which we mentioned in the hind itself.
So, option A itself is the right answer.
But it is also a duty to check remaining options too. Let us have a look on to those.
Another option mentioned above is the Lavoisier experiment. His experiments deal with the study of chemical characters of different gases. He also dealt with the burning of phosphorus and sulfur for the experiment. No such cases are observed in the above diagram. So we can clearly say it is not a Lavoisier experiment.
Also another option mentioned above is the Blaise Pascal Carries . This method actually defines Pascal's calculator. His calculator was successful in the design of its carry mechanism in which it adds one to nine on one dial, and carries one to the next dial when the first dial changes from nine to zero. No such calculator can be observed in the above diagram. So it is sure to know that this is not the right answer from the diagram itself on a first outlook.
These two options are entirely different from the option which is a fountain experiment and shows no similarity of its components with the above diagram.
Hence option A is correct.
Note: The main aim of this experiment was to show the high solubility of hydrochloric gas in water. It also proved the hydrochloric gas is acidic in nature as it turned the blue litmus paper red.
This experiment was important in understanding the features of hydrochloric gas.
Complete answer:
At first we have to look into the fountain experiment.
This method is done by introducing water through an inlet to a container with hydrogen chloride gas.
Then the gas gets mixed with water and we can see gradually the pressure inside the container drops down.
Then more water is forced to enter the container from another inlet producing a fountain effect.
This experiment is used to introduce concepts like solubility and gas laws.
Then blue litmus is added to check the acidic nature of hydrogen chloride gas in the round bottom flask.
From the definition of this experiment, it clearly defines the highlighted components like hydrogen chloride, blue litmus which we mentioned in the hind itself.
So, option A itself is the right answer.
But it is also a duty to check remaining options too. Let us have a look on to those.
Another option mentioned above is the Lavoisier experiment. His experiments deal with the study of chemical characters of different gases. He also dealt with the burning of phosphorus and sulfur for the experiment. No such cases are observed in the above diagram. So we can clearly say it is not a Lavoisier experiment.
Also another option mentioned above is the Blaise Pascal Carries . This method actually defines Pascal's calculator. His calculator was successful in the design of its carry mechanism in which it adds one to nine on one dial, and carries one to the next dial when the first dial changes from nine to zero. No such calculator can be observed in the above diagram. So it is sure to know that this is not the right answer from the diagram itself on a first outlook.
These two options are entirely different from the option which is a fountain experiment and shows no similarity of its components with the above diagram.
Hence option A is correct.
Note: The main aim of this experiment was to show the high solubility of hydrochloric gas in water. It also proved the hydrochloric gas is acidic in nature as it turned the blue litmus paper red.
This experiment was important in understanding the features of hydrochloric gas.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




