
Phenol is more acidic than that of ethanol because:
(A). Phenoxide ion is bulkier than ethoxide.
(B). Phenoxide ion is a stronger base than ethoxide.
(C). Phenoxide ion is stabilized through delocalisation.
(D). Phenoxide ion is less stable than ethoxide.
Answer
604.8k+ views
- Hint: The acidic character depends on the stability of the ion which is left behind after ${ H }^{ + }$ is released. Phenoxide ion is aromatic and can undergo resonance but ethoxide ion can’t.
Complete step-by-step solution -
Phenol is more acidic than that of ethanol because phenoxide ion is stabilized through delocalisation. Let us understand the reason why phenol is more acidic than ethanol.
The chemical formula of phenol is ${ C }_{ 6 }{ H }_{ 5 }-OH$. An acid loses ${ H }^{ + }$ ions in water. When phenol loses an ${ H }^{ + }$ ion, the ion formed is known as phenoxide ion. Its chemical formula is ${ C }_{ 6 }{ H }_{ 5 }-{ O}^{ -}$.
The negative charge formed as a result of losing an ${ H }^{ + }$ ion is not localised in phenol. The phenyl ring ${ C }_{ 6 }{ H }_{ 5 }-$ is an aromatic ring and it helps in delocalisation of the negative charge inside the ring.
This effect is called the resonance effect. It is the withdrawn or releasing effect of electrons from a particular substituent through the delocalisation of a charge.
The resonance effect takes place in compounds having a conjugated double bond system. In the conjugated double bond system, there are alternate single and double bonds. The phenoxide ion has an alternate system of single and double bonds. The structure of the phenoxide ion is given below.
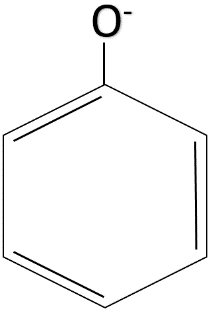
The resonance structures which make the phenoxide ion stable are given below.
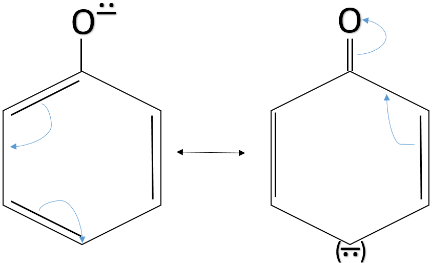
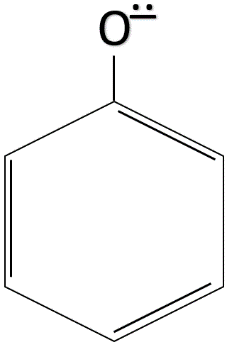
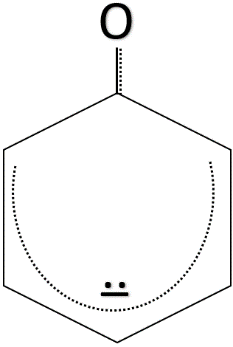


The chemical formula of ethanol is ${ CH }_{ 3 }-{ CH }_{ 2 }-OH$. As we can see that ethanol does not have any conjugated double bond system. So, when the ${ H }^{ + }$ ion leaves ethanol, the ethoxide ion formed is not stable as no resonance takes place and the negative charge is not delocalised. The formula of ethoxide ion is ${ CH }_{ 3 }-{ CH }_{ 2 }-{ O }^{ - }$.
Note: Always remember that in resonance effect the position of individual atoms does not move at all. Only the electrons in the pi cloud (Double bonded electrons) can be moved. Don’t confuse yourself while drawing resonance structures.
Complete step-by-step solution -
Phenol is more acidic than that of ethanol because phenoxide ion is stabilized through delocalisation. Let us understand the reason why phenol is more acidic than ethanol.
The chemical formula of phenol is ${ C }_{ 6 }{ H }_{ 5 }-OH$. An acid loses ${ H }^{ + }$ ions in water. When phenol loses an ${ H }^{ + }$ ion, the ion formed is known as phenoxide ion. Its chemical formula is ${ C }_{ 6 }{ H }_{ 5 }-{ O}^{ -}$.
The negative charge formed as a result of losing an ${ H }^{ + }$ ion is not localised in phenol. The phenyl ring ${ C }_{ 6 }{ H }_{ 5 }-$ is an aromatic ring and it helps in delocalisation of the negative charge inside the ring.
This effect is called the resonance effect. It is the withdrawn or releasing effect of electrons from a particular substituent through the delocalisation of a charge.
The resonance effect takes place in compounds having a conjugated double bond system. In the conjugated double bond system, there are alternate single and double bonds. The phenoxide ion has an alternate system of single and double bonds. The structure of the phenoxide ion is given below.

The resonance structures which make the phenoxide ion stable are given below.





The chemical formula of ethanol is ${ CH }_{ 3 }-{ CH }_{ 2 }-OH$. As we can see that ethanol does not have any conjugated double bond system. So, when the ${ H }^{ + }$ ion leaves ethanol, the ethoxide ion formed is not stable as no resonance takes place and the negative charge is not delocalised. The formula of ethoxide ion is ${ CH }_{ 3 }-{ CH }_{ 2 }-{ O }^{ - }$.
Note: Always remember that in resonance effect the position of individual atoms does not move at all. Only the electrons in the pi cloud (Double bonded electrons) can be moved. Don’t confuse yourself while drawing resonance structures.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




