
Phenol is obtained from cumene by the use of:
a.) Superoxide
b.) Suboxide
c.) Peroxide
d.) Sesquioxide
Answer
584.7k+ views
Hint: Cumene is an organic compound, in which an isopropyl group is attached to a benzene ring. Phenol is a compound that has a hydroxyl group attached to benzene. Therefore, cumene needs to be oxidized for converting it to phenol.
Complete step by step answer:
Cumene is prepared by Friedel-Crafts alkylation of benzene with propylene in presence of anhydrous aluminium chloride.
The conversion of cumene to phenol takes place in the following manner –
Step 1 –
Cumene is oxidized in the presence of air to form an intermediate structure called cumene hydroperoxide. This takes place in an alkaline medium.
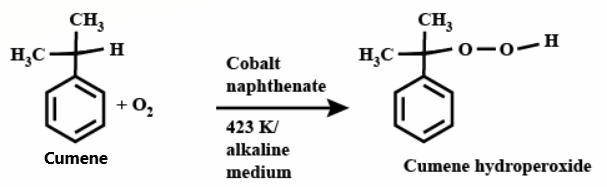
Cumene hydroperoxide is then treated with a dilute acid in the presence of heat, which cleaves the compound via Hock rearrangement to form phenol and acetone.
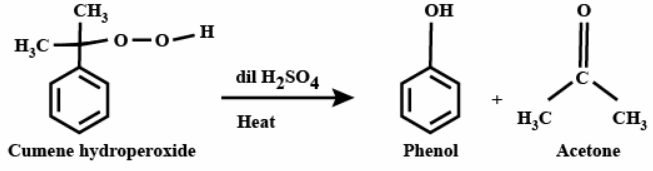
As we can see, the intermediate compound formed was cumene hydroperoxide. Therefore, phenol is obtained from cumene by the use of peroxide.
So, the correct answer is “Option C”.
Note: The IUPAC name of cumene is isopropyl benzene.
This process, for the conversion of cumene to phenol is also known as ‘Hock process’. It is a widely used reaction, for synthesizing acetone. The process starts by preparing cumene from benzene and propylene, which then goes through the process given above to give phenol and acetone respectively.
Complete step by step answer:
Cumene is prepared by Friedel-Crafts alkylation of benzene with propylene in presence of anhydrous aluminium chloride.
The conversion of cumene to phenol takes place in the following manner –
Step 1 –
Cumene is oxidized in the presence of air to form an intermediate structure called cumene hydroperoxide. This takes place in an alkaline medium.

Cumene hydroperoxide is then treated with a dilute acid in the presence of heat, which cleaves the compound via Hock rearrangement to form phenol and acetone.

As we can see, the intermediate compound formed was cumene hydroperoxide. Therefore, phenol is obtained from cumene by the use of peroxide.
So, the correct answer is “Option C”.
Note: The IUPAC name of cumene is isopropyl benzene.
This process, for the conversion of cumene to phenol is also known as ‘Hock process’. It is a widely used reaction, for synthesizing acetone. The process starts by preparing cumene from benzene and propylene, which then goes through the process given above to give phenol and acetone respectively.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




