
Sucrose on hydrolysis gives?
(A) Glucose + Glucose
(B) Glucose + Galactose
(C) Glucose + Fructose
(D) Glucose + Lactose
Answer
582.9k+ views
Hint: Sucrose is a disaccharide made by the reaction between two aldohexose. Draw the expanded structure of sucrose. Now try to split the complex structure into two monosaccharides such that both compounds have 6 carbon atoms. Now identify the compounds obtained and then write the IUPAC name for the same.
Complete step by step answer:
Sucrose is considered as common sugar. Sucrose is produced naturally in plants from which the edible table sugar is refined. The molecular formula for sucrose is ${{C}_{12}}{{H}_{22}}{{O}_{11}}$. We will now draw the expanded structure of a sucrose molecule.
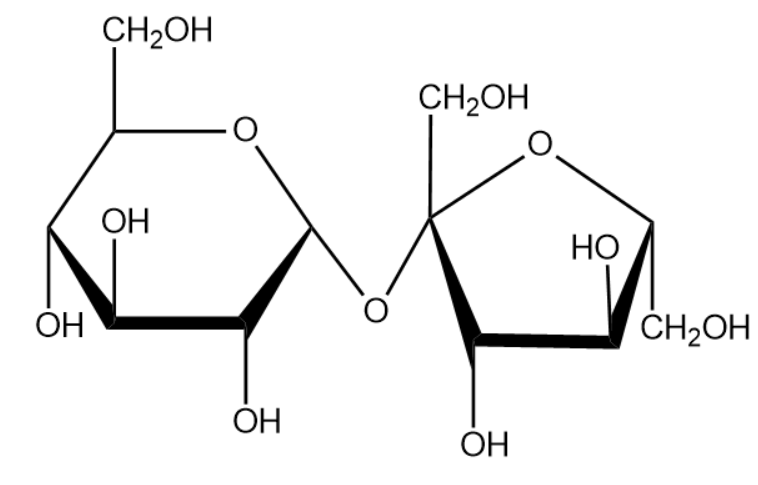
From the above structure we can identify the monosaccharide that help to from the compound sucrose. The structure compounds are given below:
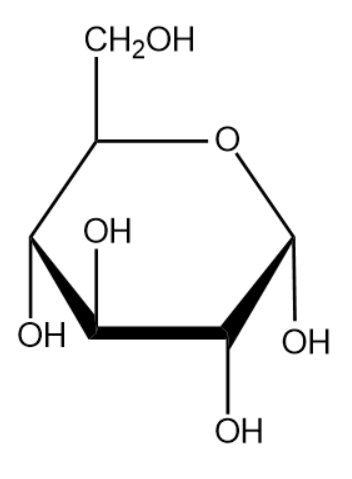
The name of the above monosaccharide is glucose.
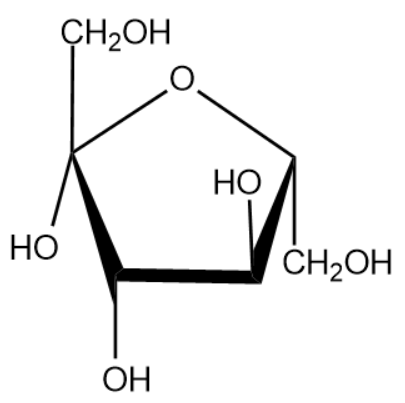
The name of the above monosaccharide is fructose.
From the above statements we can conclude that sucrose on hydrolysis will give the respective monosaccharides, glucose and fructose. So, the correct answer is “Option C”.
Note: It is important to know that sucrose is dextrorotatory in nature. However, upon hydrolysis the products obtained are dextrorotatory glucose and laevorotatory fructose. Since the rotation of fructose is much more than that to glucose, the mixture as a whole is considered laevorotatory. Since, the hydrolysis of sucrose brings a significant change in the type of rotation to the mixture, i.e. from dextrorotatory to laevorotatory, it is also called as invert sugar.
Complete step by step answer:
Sucrose is considered as common sugar. Sucrose is produced naturally in plants from which the edible table sugar is refined. The molecular formula for sucrose is ${{C}_{12}}{{H}_{22}}{{O}_{11}}$. We will now draw the expanded structure of a sucrose molecule.

From the above structure we can identify the monosaccharide that help to from the compound sucrose. The structure compounds are given below:

The name of the above monosaccharide is glucose.

The name of the above monosaccharide is fructose.
From the above statements we can conclude that sucrose on hydrolysis will give the respective monosaccharides, glucose and fructose. So, the correct answer is “Option C”.
Note: It is important to know that sucrose is dextrorotatory in nature. However, upon hydrolysis the products obtained are dextrorotatory glucose and laevorotatory fructose. Since the rotation of fructose is much more than that to glucose, the mixture as a whole is considered laevorotatory. Since, the hydrolysis of sucrose brings a significant change in the type of rotation to the mixture, i.e. from dextrorotatory to laevorotatory, it is also called as invert sugar.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE

In a human foetus the limbs and digits develop after class 12 biology CBSE

AABbCc genotype forms how many types of gametes a 4 class 12 biology CBSE

The correct structure of ethylenediaminetetraacetic class 12 chemistry CBSE




