
The central oxygen atom in ether is ----
Answer
508.8k+ views
Hint: In ether molecules one atom of oxygen is combined with two groups of alkyl or aryl groups. On the basis of the nature of substituents adjacent to oxygen atoms they are categorized as symmetrical ether or unsymmetrical ether.
Complete answer:
Electronic configuration of oxygen is $1{s^2}2{s^2}2{p_x}^22{p_y}^12{p_z}^1$ and having two unpaired electrons present in its p-orbital.
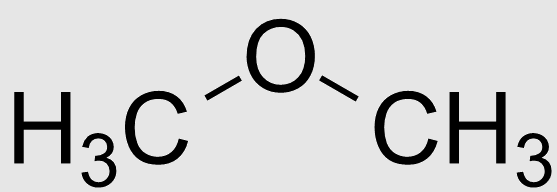
Let the ether compound is dimethyl ether with molecular formula $C{H_3}OC{H_3}$
In ether moiety central oxygen forms a bond with the carbon atom of alkyl group.
Four valence orbital of oxygen atom $\left[ {2s,2{p_x},2{p_y},2{p_z}} \right]$ undergoes hybridization to form four orbitals of equal energy $\left( {s{p^3}} \right)$.
Two hydrogen atoms of the alkyl group form a bond with $\left[ {2{p_y}^1,2{p_z}^1} \right]$ to form a sigma bond .
Hence two lone pair electrons are still present in the oxygen atom in its $\left[ {2{s^2},2{p^2}} \right]$ orbitals.
These two lone pairs arrange themselves at a particular angle to form a stable structure.
These lone pair pushed the bond between oxygen and hydrogen $\left( {O - H} \right)$ toward each other and reduces the bond angle to $\left( {{{104.5}^ \circ }} \right)$ and acquired the final shape as Bent or V-shaped molecule.
Hence from the above discussion we see that the central oxygen atom in ether is $s{p^3}$ hybridized with two lone pairs of electrons.
Note:
Other molecules shows $s{p^3}$ hybridization includes- $C{H_4},SnC{l_4},SiC{l_4},Si{F_4},N{H_4}^ + $ etc.
Regular geometry of $s{p^3}$ hybridization is tetrahedral, but it may vary due the presence of lone pairs of electrons in the compound.
Complete answer:
Electronic configuration of oxygen is $1{s^2}2{s^2}2{p_x}^22{p_y}^12{p_z}^1$ and having two unpaired electrons present in its p-orbital.
Let the ether compound is dimethyl ether with molecular formula $C{H_3}OC{H_3}$
In ether moiety central oxygen forms a bond with the carbon atom of alkyl group.
Four valence orbital of oxygen atom $\left[ {2s,2{p_x},2{p_y},2{p_z}} \right]$ undergoes hybridization to form four orbitals of equal energy $\left( {s{p^3}} \right)$.
Two hydrogen atoms of the alkyl group form a bond with $\left[ {2{p_y}^1,2{p_z}^1} \right]$ to form a sigma bond .
Hence two lone pair electrons are still present in the oxygen atom in its $\left[ {2{s^2},2{p^2}} \right]$ orbitals.
These two lone pairs arrange themselves at a particular angle to form a stable structure.
These lone pair pushed the bond between oxygen and hydrogen $\left( {O - H} \right)$ toward each other and reduces the bond angle to $\left( {{{104.5}^ \circ }} \right)$ and acquired the final shape as Bent or V-shaped molecule.
Hence from the above discussion we see that the central oxygen atom in ether is $s{p^3}$ hybridized with two lone pairs of electrons.

Note:
Other molecules shows $s{p^3}$ hybridization includes- $C{H_4},SnC{l_4},SiC{l_4},Si{F_4},N{H_4}^ + $ etc.
Regular geometry of $s{p^3}$ hybridization is tetrahedral, but it may vary due the presence of lone pairs of electrons in the compound.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

10 examples of friction in our daily life




