
The correct electron dot structure of water molecule is :
(A)

(B)
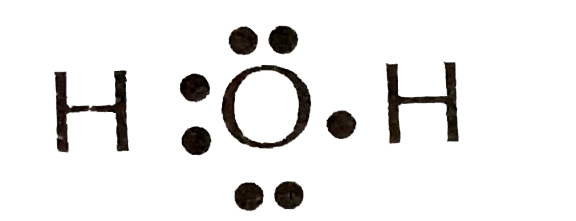
(C)

(D)

Answer
529.6k+ views
Hint: The atomic number of oxygen is 8. So, we can say that there are 6 valence electrons. And the atomic number of hydrogen is 1. So, the number of valence electrons is 1.
Complete step by step answer:
The electronic configuration of oxygen is 2, 6. So there are 6 valence electrons available for oxygen in order to form a chemical bonding. Similarly, the electronic configuration of hydrogen is 1. So, there is 1 valence electron available for hydrogen in order to form a chemical bonding.
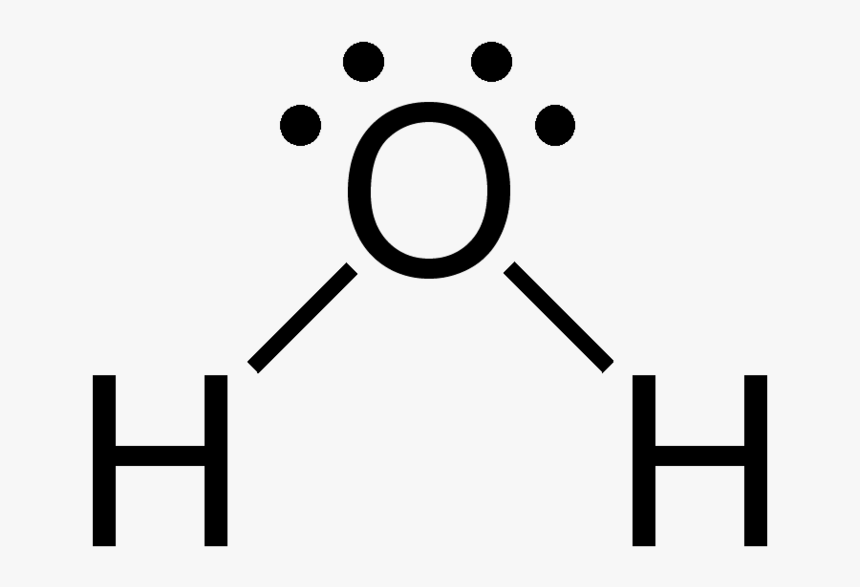
In the diagram, provided above we get an idea about the structure of water molecules.
Oxygen atom has 6 valence electrons out of which 2 valence electrons are involved in bonding with the 2 hydrogen atoms. Therefore, a water molecule has 2 bond pairs of electrons and 2 lone pair (non-bonded) pairs of electrons.
So, we can say that the Lewis dot structure of of water molecule is:

Therefore, corresponding to the question, we can that the correct Lewis dot structure of water molecules is number 3.
So, Option C is the correct answer.
Note:
(1) Lewis structures also known as Lewis dot structures or electron dot structures are diagrams that represent the valence electrons of an atom within a molecule. These Lewis symbols are Lewis structures which help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds.
(2) In order to draw the Lewis dot structure, place any remaining lone electron pairs on the central atom. Usually, electronegative atoms will have lone pairs on them. The number of electrons placed on the molecule should correspond to the total number of valence electrons of the atoms in the molecule.
Complete step by step answer:
The electronic configuration of oxygen is 2, 6. So there are 6 valence electrons available for oxygen in order to form a chemical bonding. Similarly, the electronic configuration of hydrogen is 1. So, there is 1 valence electron available for hydrogen in order to form a chemical bonding.

In the diagram, provided above we get an idea about the structure of water molecules.
Oxygen atom has 6 valence electrons out of which 2 valence electrons are involved in bonding with the 2 hydrogen atoms. Therefore, a water molecule has 2 bond pairs of electrons and 2 lone pair (non-bonded) pairs of electrons.
So, we can say that the Lewis dot structure of of water molecule is:

Therefore, corresponding to the question, we can that the correct Lewis dot structure of water molecules is number 3.
So, Option C is the correct answer.
Note:
(1) Lewis structures also known as Lewis dot structures or electron dot structures are diagrams that represent the valence electrons of an atom within a molecule. These Lewis symbols are Lewis structures which help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds.
(2) In order to draw the Lewis dot structure, place any remaining lone electron pairs on the central atom. Usually, electronegative atoms will have lone pairs on them. The number of electrons placed on the molecule should correspond to the total number of valence electrons of the atoms in the molecule.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

Hydrocarbons Class 11 Chemistry Chapter 9 CBSE Notes - 2025-26

Thermodynamics Class 11 Chemistry Chapter 5 CBSE Notes - 2025-26

Equilibrium Class 11 Chemistry Chapter 6 CBSE Notes - 2025-26

Organic Chemistry Some Basic Principles And Techniques Class 11 Chemistry Chapter 8 CBSE Notes - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 7 Redox Reactions (2025-26)




