
The correct structural formula of butanoic acid is:
A.
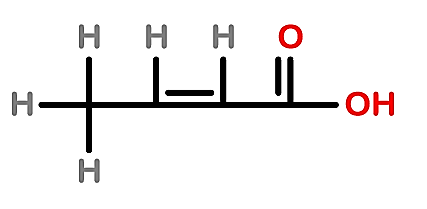
B.
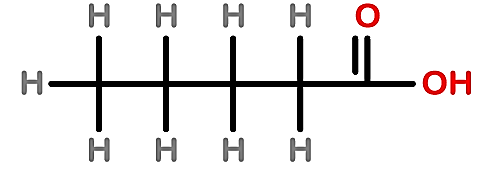
C.
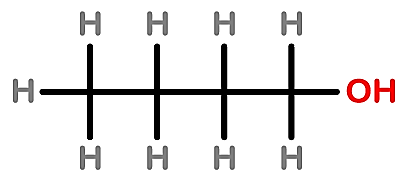
D.
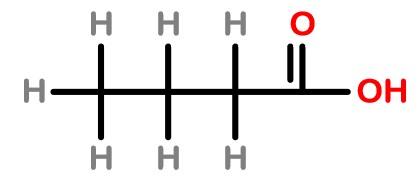




Answer
583.2k+ views
Hint: For this problem, we know that the word 'but' is used when there are for carbon atoms present in the main chain. Similarly, we have to explain about the prefix 'oic acid' so that we can choose the correct options.
Complete step by step answer:
- In the given question, we have to choose the correct structure of butanoic acid among the given options.
- As we know that the different functional groups have a different prefix that is used for writing the IUPAC name of the compound.
- Now, we know that the prefix 'but' is used when there are four carbon atoms present in the chain and the prefix 'oic acid' is used when the functional group carboxylic acid is used.
- Now, the carboxylic acid is considered as the prior group that's why the prefix oic acid is used while doing the IUPAC naming whereas if another functional group which is more prior than it then the suffix 'carboxy' is used.
- Whereas there is no position mentioned of the carboxylic acid in the IUPAC name which means that the group is present on the first carbon.
- So, the structure 4 will be the correct structural formula of butanoic acid because there are four carbon atoms in which carboxylic acid is present on the first carbon.
- Whereas in 1 structure double bond is present on the second carbon so the IUPAC name of the compound will be but - 2 - ene - 1 - oic acid.
So, the correct answer is “Option D”.
Note: In 2 structure, there are 5 carbons present in the long-chain so this option is also incorrect whereas, in 3 structure, there is no carboxylic acid present instead of it alcohol group is present so the prefix 'ol' will be used.
Complete step by step answer:
- In the given question, we have to choose the correct structure of butanoic acid among the given options.
- As we know that the different functional groups have a different prefix that is used for writing the IUPAC name of the compound.
- Now, we know that the prefix 'but' is used when there are four carbon atoms present in the chain and the prefix 'oic acid' is used when the functional group carboxylic acid is used.
- Now, the carboxylic acid is considered as the prior group that's why the prefix oic acid is used while doing the IUPAC naming whereas if another functional group which is more prior than it then the suffix 'carboxy' is used.
- Whereas there is no position mentioned of the carboxylic acid in the IUPAC name which means that the group is present on the first carbon.
- So, the structure 4 will be the correct structural formula of butanoic acid because there are four carbon atoms in which carboxylic acid is present on the first carbon.
- Whereas in 1 structure double bond is present on the second carbon so the IUPAC name of the compound will be but - 2 - ene - 1 - oic acid.
So, the correct answer is “Option D”.
Note: In 2 structure, there are 5 carbons present in the long-chain so this option is also incorrect whereas, in 3 structure, there is no carboxylic acid present instead of it alcohol group is present so the prefix 'ol' will be used.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




