
The IUPAC name of the compound shown below is
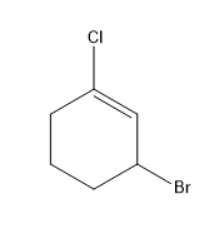
(A) $ 2 - Bromo - 6 - chlorocyclohex - 1 - ene $
(B) $ 6 - bromo - 2 - chlorocyclohexene $
(C) $ 3 - Bromo - 1 - chlorocyclohex - 1 - ene $
(D) $ 1 - bromo - 3 - chlorocyclohexene $

Answer
483.6k+ views
Hint: Cycloalkenes are the alkenes arranged in cyclic form. When the substituents like two different halogens were present on the cycloalkene, then the substituents must be written in the alphabetical order only and the numbering of substituents must get the lowest number.
Complete answer:
Chemical compounds were simply classified into functional groups based on the groups present in it. hydrocarbons are chemical compounds consisting of only carbon and hydrogen atoms. Alkenes are unsaturated hydrocarbons consisting of one or more carbon-carbon double-bonded atoms.
Cycloalkenes are alkenes that are arranged in a cyclic form. In the given structure, there is a cyclohexene group. As the ring consists of six carbon atoms, in addition to this cycloalkene ring there is a presence of two different halogens which are chlorine and bromine.
The numbering should be given to the above structure, as the double-bonded carbon gets the lowest number. As there were two substituents the substituent which comes first in alphabetical order should be written first. Thus, bromine will be written first and the numbering should start from the double-bonded carbon.

Thus, the IUPAC name of the given structure is $ 3 - Bromo - 1 - chlorocyclohex - 1 - ene $
Option C is the correct one.
Note:
In the given structure, both chlorine and the double-bonded carbon get the same number. Unlike this, when the numbering starts from another double-bonded carbon, the substituents will not get the lowest number. Thus, the above order must be considered while writing the IUPAC nomenclature.
Complete answer:
Chemical compounds were simply classified into functional groups based on the groups present in it. hydrocarbons are chemical compounds consisting of only carbon and hydrogen atoms. Alkenes are unsaturated hydrocarbons consisting of one or more carbon-carbon double-bonded atoms.
Cycloalkenes are alkenes that are arranged in a cyclic form. In the given structure, there is a cyclohexene group. As the ring consists of six carbon atoms, in addition to this cycloalkene ring there is a presence of two different halogens which are chlorine and bromine.
The numbering should be given to the above structure, as the double-bonded carbon gets the lowest number. As there were two substituents the substituent which comes first in alphabetical order should be written first. Thus, bromine will be written first and the numbering should start from the double-bonded carbon.

Thus, the IUPAC name of the given structure is $ 3 - Bromo - 1 - chlorocyclohex - 1 - ene $
Option C is the correct one.
Note:
In the given structure, both chlorine and the double-bonded carbon get the same number. Unlike this, when the numbering starts from another double-bonded carbon, the substituents will not get the lowest number. Thus, the above order must be considered while writing the IUPAC nomenclature.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

10 examples of friction in our daily life




