
Which element comes below Sulphur in the periodic table?
A)Selenium
B)Oxygen
C)Bromine
D)Germanium
Answer
585.3k+ views
Hint: Basically, Sulphur is found in group 16 in the periodic table. It is a non-metal and is obtained as a byproduct after the production of natural gas. It is bright yellow in color and has an extremely bad odor (like rotten eggs).
Complete step by step answer:
Sulphur is a chemical element with the symbol S and atomic number 16. It is basically a bright yellow crystalline solid and under normal conditions its atoms form cyclic octatomic molecules with the chemical formula ${S_8}$ .
Moreover, this element is found in group 16 in the periodic table and among the given options the element that comes just below Sulphur is Selenium. The periodic table is as shown:
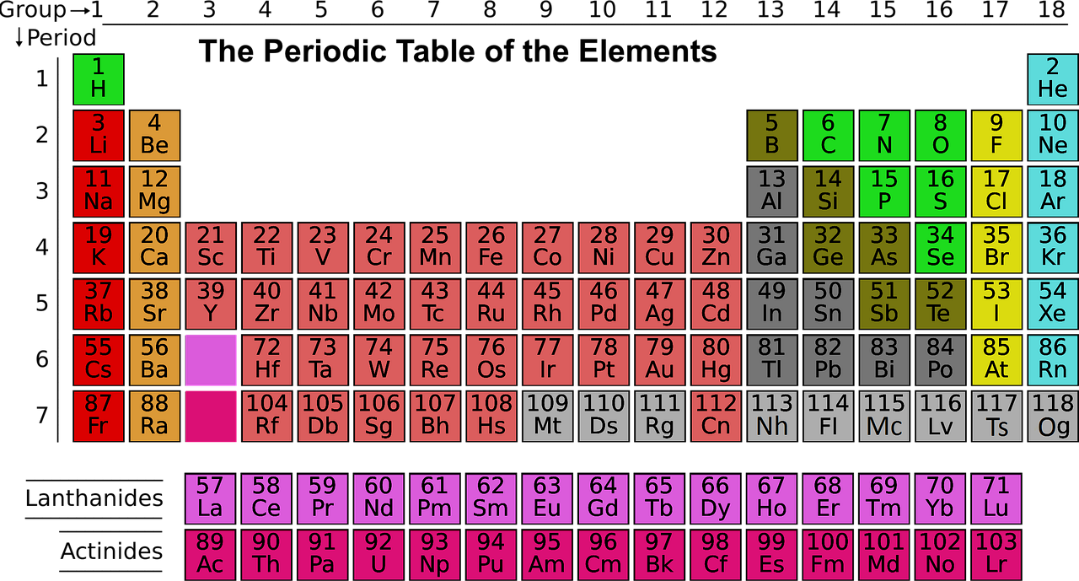
Now, Selenium is often found in the combination with metals like mercury, copper, silver or lead. It is also a member of group 16 of the periodic table. This element is rare and is composed of 90 parts per billion of the Earth’s crust. It basically appears in different allotropic forms such as red amorphous powder, gray crystalline metal and a red crystalline material known as metallic selenium.
Hence, Option A is correct.
Note:Selenium often serves as a decolorizer when incorporated in small amounts into the glass. Moreover, when used in large quantity, it imparts a clear red color to the glass. It is also used in the preparation of read enamels for steel and ceramics and most of the metals are generally insoluble in this element.
Complete step by step answer:
Sulphur is a chemical element with the symbol S and atomic number 16. It is basically a bright yellow crystalline solid and under normal conditions its atoms form cyclic octatomic molecules with the chemical formula ${S_8}$ .
Moreover, this element is found in group 16 in the periodic table and among the given options the element that comes just below Sulphur is Selenium. The periodic table is as shown:

Now, Selenium is often found in the combination with metals like mercury, copper, silver or lead. It is also a member of group 16 of the periodic table. This element is rare and is composed of 90 parts per billion of the Earth’s crust. It basically appears in different allotropic forms such as red amorphous powder, gray crystalline metal and a red crystalline material known as metallic selenium.
Hence, Option A is correct.
Note:Selenium often serves as a decolorizer when incorporated in small amounts into the glass. Moreover, when used in large quantity, it imparts a clear red color to the glass. It is also used in the preparation of read enamels for steel and ceramics and most of the metals are generally insoluble in this element.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




