
Which reaction give cyclic ketone as a product?
A.
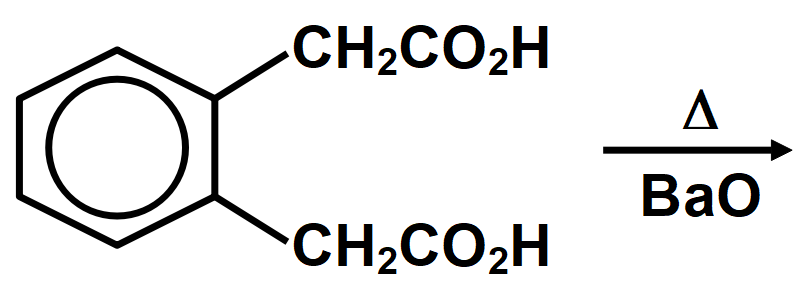
B.
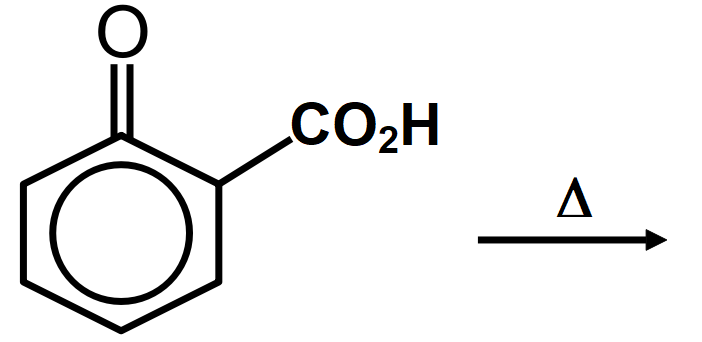
C.
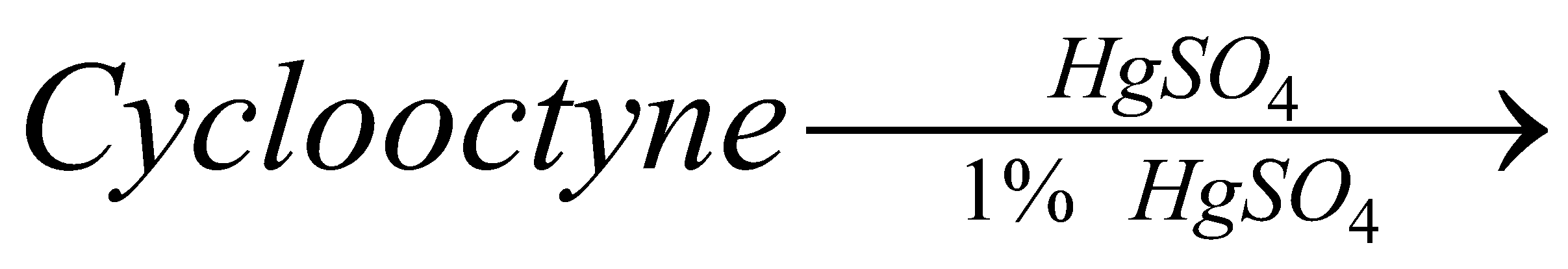
D.
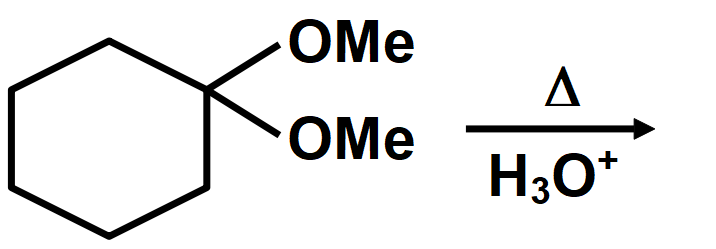
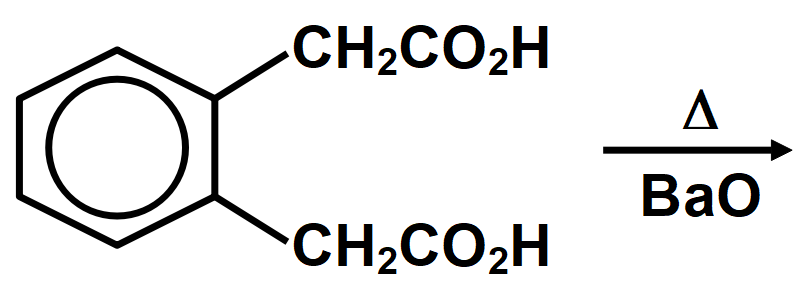

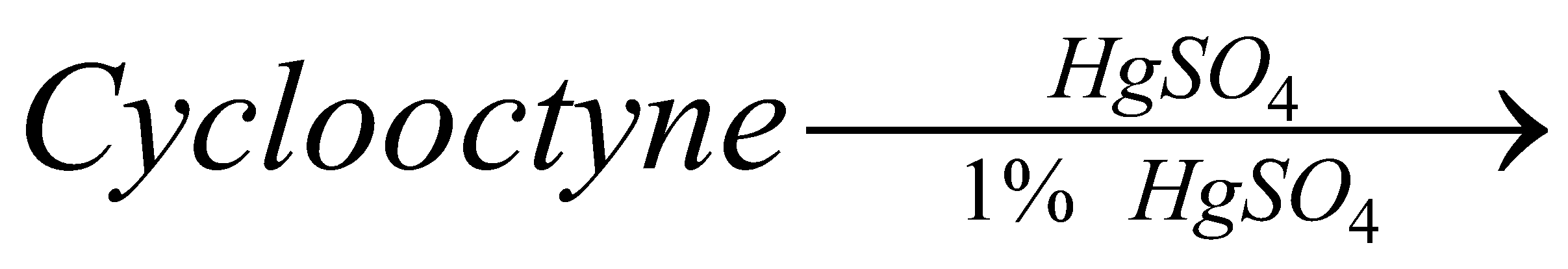
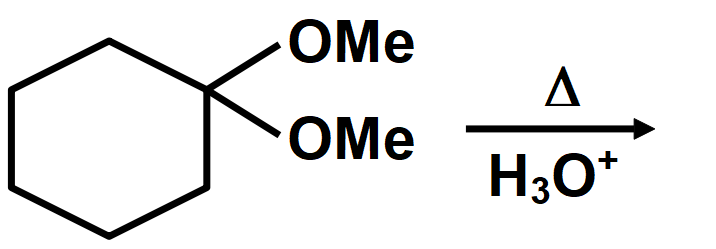
Answer
509.1k+ views
Hint: We know that the cyclic acetals are generally formed when the reaction between the two molecules i.e. a ketone and a diol take place. This reaction results in the formation of two main products i.e. a ketal and water.
Complete answer:
Cyclic acetals or cyclic acetals are more stable in comparison to acetals because of the presence of chelate effect that is majorly derived from having both groups of acetals being connected to each other in diol. Cyclic acetals have their major applications in carbohydrate chemistry. alkyl groups are close enough then we have the possibility of them interacting with each other and these reactions occur when the dicarboxylic are heated. When dicarboxylic acids are heated, they tend to undergo cyclization to form cyclic ketone. But for cyclization to happen the ring formed should not have strain which means that five and six membered rings are the most favored because of their least angle strain. Now in our question the first compound cannot undergo cyclization because a ring of two carbon atoms is not possible. Compound A which on cyclization will give a six membered ring which is stable, so this compound is capable of undergoing cyclization and will give cyclic ketone as shown below:
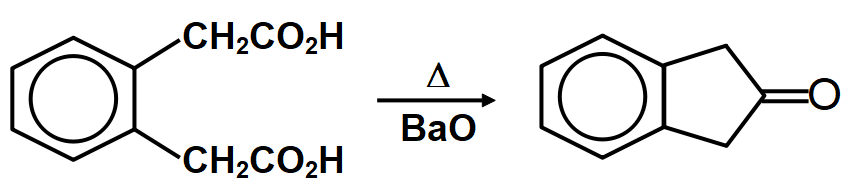
Therefore, the correct answer is option A.
Note:
Remember that a ketal refers to a functional group that has been derived from a ketone with the replacement of a carbonyl $\left( C=O \right)$ group by two alkoxy groups. In other words, a ketal can also be referred to as an acetal derived from the ketone. Therefore, a cyclic ketal is a ketal in which ketal carbon along with one or both oxygen atoms are members of a ring.
Complete answer:
Cyclic acetals or cyclic acetals are more stable in comparison to acetals because of the presence of chelate effect that is majorly derived from having both groups of acetals being connected to each other in diol. Cyclic acetals have their major applications in carbohydrate chemistry. alkyl groups are close enough then we have the possibility of them interacting with each other and these reactions occur when the dicarboxylic are heated. When dicarboxylic acids are heated, they tend to undergo cyclization to form cyclic ketone. But for cyclization to happen the ring formed should not have strain which means that five and six membered rings are the most favored because of their least angle strain. Now in our question the first compound cannot undergo cyclization because a ring of two carbon atoms is not possible. Compound A which on cyclization will give a six membered ring which is stable, so this compound is capable of undergoing cyclization and will give cyclic ketone as shown below:

Therefore, the correct answer is option A.
Note:
Remember that a ketal refers to a functional group that has been derived from a ketone with the replacement of a carbonyl $\left( C=O \right)$ group by two alkoxy groups. In other words, a ketal can also be referred to as an acetal derived from the ketone. Therefore, a cyclic ketal is a ketal in which ketal carbon along with one or both oxygen atoms are members of a ring.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




