
Write the equation for the preparation of acetanilide from aniline?
Answer
584.7k+ views
Hint: In the synthesis of acetanilide, pure distilled aniline was used to get a good amount of yield and a small amount of zinc can be added to the mixture, which reduces coloured impurities inline, also prevents oxidation while reaction continues. It has been used as an intermediate in rubber accelerator synthesis and dye synthesis.
Complete step by step solution:
N-acetyl arylamine is an odourless solid chemical of leaf or flake-like appearance. It is known as acetanilide, which is a white organic solid compound used in organic synthesis. N-phenylacetamide, acetanilide, are other names of the compound.
Chemicals required: aniline, acetic anhydride, and zinc dust.
Principle: acetanilide synthesis from aniline by acetylating it with acetic anhydride in the presence of glacial acetic acid. Aniline which is primary amine was basic. Acetic anhydride, an anhydride of acetic acid, acts here as a source of an acyl group.
(1) The preparation of acetanilide from aniline has belonged to a nucleophilic substitution reaction. Acetic anhydride is prepared by fractional distillation by reaction with sodium acetate and acetyl chloride.
(2) In this reaction, aniline acts as a nucleophile, and the acyl (\[C{{H}_{3}}CO-\] ) group from acetic anhydride acts as an electrophile. Here the hydrogen atom of $-N{{H}_{2}}$, the group is replaced by the acyl group.
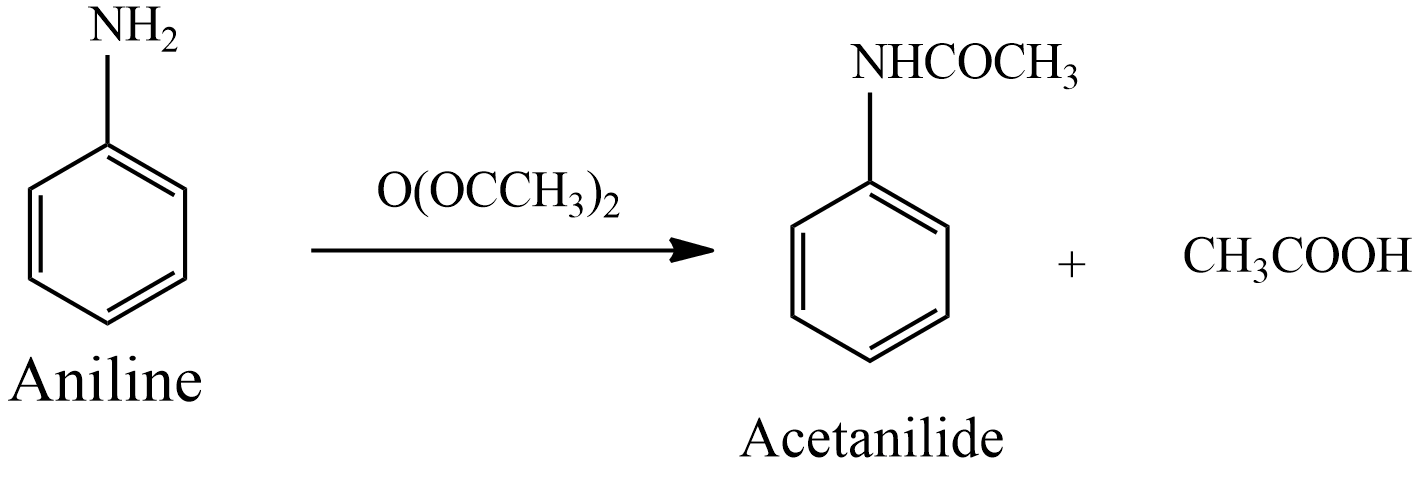
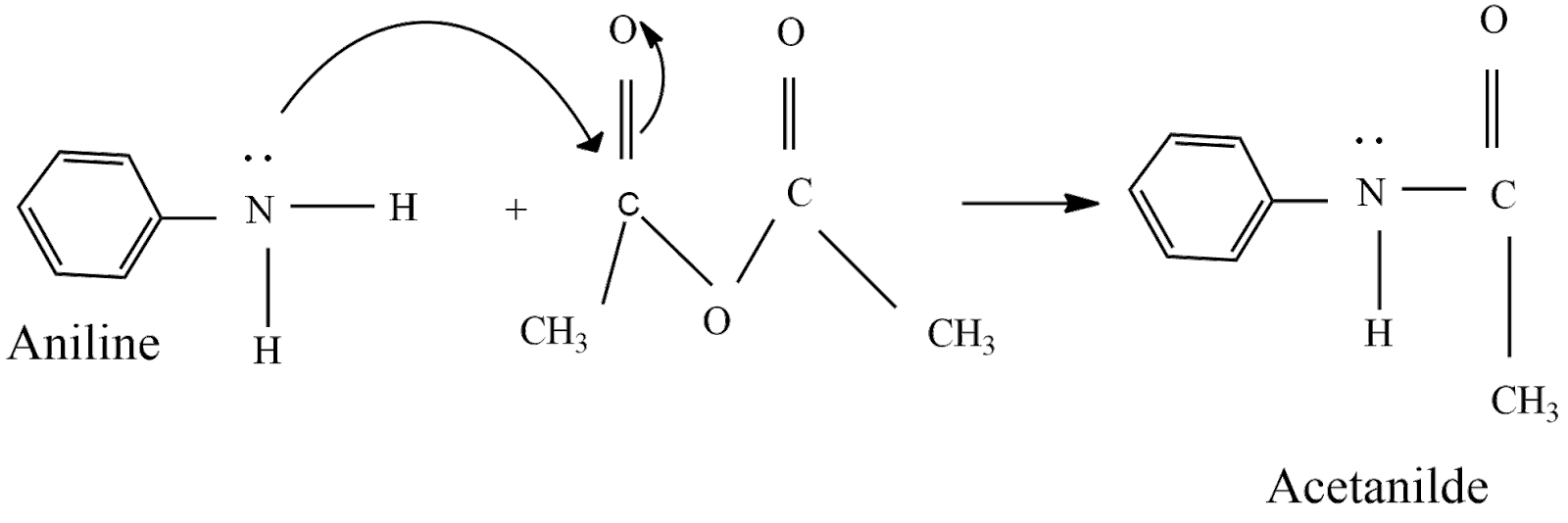
Hence, The above reaction shows the equation of preparation of acetanilide from aniline.
Note: If this synthesis is done in the lab, Acetanilide is a carcinogenic compound, which can cause cancer in humans. Don’t inhale the fumes of acetic anhydride during its preparation by fractional distillation. Vigorous heating and keep stirring the mixture added by ice-cold water. Acetanilide is a polar compound because of the amide group. This was used in the past to treat fever and headache and was known as Antifebrin by its brand name.
Complete step by step solution:
N-acetyl arylamine is an odourless solid chemical of leaf or flake-like appearance. It is known as acetanilide, which is a white organic solid compound used in organic synthesis. N-phenylacetamide, acetanilide, are other names of the compound.
Chemicals required: aniline, acetic anhydride, and zinc dust.
Principle: acetanilide synthesis from aniline by acetylating it with acetic anhydride in the presence of glacial acetic acid. Aniline which is primary amine was basic. Acetic anhydride, an anhydride of acetic acid, acts here as a source of an acyl group.
(1) The preparation of acetanilide from aniline has belonged to a nucleophilic substitution reaction. Acetic anhydride is prepared by fractional distillation by reaction with sodium acetate and acetyl chloride.
(2) In this reaction, aniline acts as a nucleophile, and the acyl (\[C{{H}_{3}}CO-\] ) group from acetic anhydride acts as an electrophile. Here the hydrogen atom of $-N{{H}_{2}}$, the group is replaced by the acyl group.


Hence, The above reaction shows the equation of preparation of acetanilide from aniline.
Note: If this synthesis is done in the lab, Acetanilide is a carcinogenic compound, which can cause cancer in humans. Don’t inhale the fumes of acetic anhydride during its preparation by fractional distillation. Vigorous heating and keep stirring the mixture added by ice-cold water. Acetanilide is a polar compound because of the amide group. This was used in the past to treat fever and headache and was known as Antifebrin by its brand name.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




