
Write the structure of the following organic halogen compound
\[1 - \] Chloro \[ - 4 - \] ethylcyclohexane
Answer
516k+ views
Hint: The organic halogen compounds are actually the derivative of organic compounds , in which the hydrogen atoms are replaced by the equal number of the halogen atoms. These halogen atoms can be any halogen atom like fluorine, chlorine , bromine and iodine.
Complete answer:
There are few points to keep in mind while drawing the structure.
While drawing the structure from the given IUPAC name , always keep in mind to first draw the longest carbon chain , as in case of organic compounds draw the biggest cyclic chain.
So, let’s draw the structure of \[1 - \] Chloro \[ - 4 - \] ethylcyclohexane.
Since, the halogen atom chloro i.e. chlorine comes alphabetically before than ethane , so it will be numbered first.
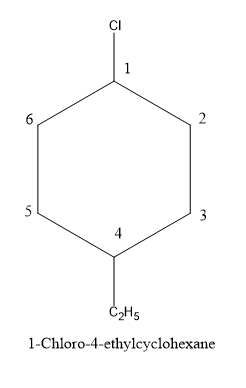
Let’s see the steps to draw the structure one by one:
First of all we will draw the structure of cyclohexane.
Then as suggested in the name i.e. \[1 - \] Chloro , so we will assign chlorine atoms at the carbon number \[1\] of the cyclohexane ring.
Further moving on , according to the name ethyl group is lying at the carbon number \[4\] of the cyclohexane ring, so we will put ethane at the carbon \[4\].
Thus, here’s our complete structure of the organic halogen compound \[1 - \] Chloro \[ - 4 - \] ethylcyclohexane.
Note:
Since , we know that all the alkyl groups, halogens , and other functional groups lying as the side chain in the compound are always named as prefixes in the nomenclature of the compounds. And the decision of which side functional group will get the first number , depends on their alphabetical order.
Complete answer:
There are few points to keep in mind while drawing the structure.
While drawing the structure from the given IUPAC name , always keep in mind to first draw the longest carbon chain , as in case of organic compounds draw the biggest cyclic chain.
So, let’s draw the structure of \[1 - \] Chloro \[ - 4 - \] ethylcyclohexane.
Since, the halogen atom chloro i.e. chlorine comes alphabetically before than ethane , so it will be numbered first.

Let’s see the steps to draw the structure one by one:
First of all we will draw the structure of cyclohexane.
Then as suggested in the name i.e. \[1 - \] Chloro , so we will assign chlorine atoms at the carbon number \[1\] of the cyclohexane ring.
Further moving on , according to the name ethyl group is lying at the carbon number \[4\] of the cyclohexane ring, so we will put ethane at the carbon \[4\].
Thus, here’s our complete structure of the organic halogen compound \[1 - \] Chloro \[ - 4 - \] ethylcyclohexane.
Note:
Since , we know that all the alkyl groups, halogens , and other functional groups lying as the side chain in the compound are always named as prefixes in the nomenclature of the compounds. And the decision of which side functional group will get the first number , depends on their alphabetical order.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




