
In electrophilic aromatic substitution reaction, the nitro group is meta directing because it:
(A) Decreases electron density at meta position
(B) Increases electron density at meta position
(C) Increases electron density at ortho and para positions
(D) Decreases electron density at ortho and para positions
Answer
233.1k+ views
Hint: Nitro group is an electron withdrawing group (EWG). When it is directly bonded to the carbon atom of benzene, it can show resonance and inductive effect which directs the position of incoming electrophile
Step-by-Step Solution:
Let us analyse this behaviour of the Nitro group in detail and see why it occurs.
- Electrophilic Aromatic Substitution (EAS) requires the benzene ring to be nucleophilic, as benzene attacks the electrophile using sites where it holds a negative charge.
- Now, when a nitro group is attached to a benzene ring, it deactivates the benzene ring towards EAS, as it prevents a negative charge from even existing in the benzene ring and makes the ring positively charged due to its electron withdrawing nature by resonance effect. How positive charge shuffles across the benzene ring is shown in the following figure by resonating structures.
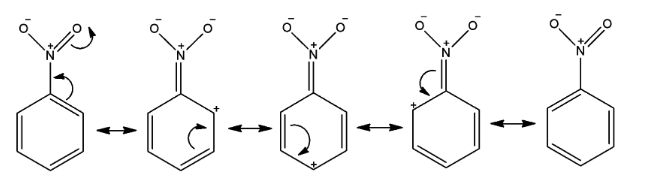
- We can see that the meta positions are less positively charged than the ortho/meta positions of the benzene ring and we need more electron density at a position at which the electrophile will connect. So, the carbon atom of meta position will attack the electrophile and as a result, a meta-substituted product will form.
- This is further proven as there is no full positive charge drawn on a meta position in the nitro group resonance forms.
- Therefore, nitro groups are meta directing, as only the meta positions can be nucleophilic enough to attack an electrophile. Remember that there is no increase in electron density at meta position.
- So, we can say that here, electron density at ortho and para position gets decreased by the nitro group and so that it is meta directing.
Note: Remember that not all EWG are meta directing. Halogens, despite being EWG, are ortho-para directors. The reason behind this is that they can donate electron density through resonance to the aromatic ring.
Step-by-Step Solution:
Let us analyse this behaviour of the Nitro group in detail and see why it occurs.
- Electrophilic Aromatic Substitution (EAS) requires the benzene ring to be nucleophilic, as benzene attacks the electrophile using sites where it holds a negative charge.
- Now, when a nitro group is attached to a benzene ring, it deactivates the benzene ring towards EAS, as it prevents a negative charge from even existing in the benzene ring and makes the ring positively charged due to its electron withdrawing nature by resonance effect. How positive charge shuffles across the benzene ring is shown in the following figure by resonating structures.

- We can see that the meta positions are less positively charged than the ortho/meta positions of the benzene ring and we need more electron density at a position at which the electrophile will connect. So, the carbon atom of meta position will attack the electrophile and as a result, a meta-substituted product will form.
- This is further proven as there is no full positive charge drawn on a meta position in the nitro group resonance forms.
- Therefore, nitro groups are meta directing, as only the meta positions can be nucleophilic enough to attack an electrophile. Remember that there is no increase in electron density at meta position.
- So, we can say that here, electron density at ortho and para position gets decreased by the nitro group and so that it is meta directing.
Note: Remember that not all EWG are meta directing. Halogens, despite being EWG, are ortho-para directors. The reason behind this is that they can donate electron density through resonance to the aromatic ring.
Recently Updated Pages
JEE Main 2026 Session 2 Registration Open, Exam Dates, Syllabus & Eligibility

JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

Trending doubts
JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Understanding Average and RMS Value in Electrical Circuits

Understanding Collisions: Types and Examples for Students

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Understanding Atomic Structure for Beginners

AssertionIn electrolytic refining of metal impure metal class 12 chemistry JEE_Main

Other Pages
JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

NCERT Solutions For Class 12 Chemistry Chapter 2 Solutions Hindi Medium (2025-26)

CBSE Class 12 Chemistry Set 1 56/2/1 2025: Question Paper, Answers & Analysis

CBSE Class 12 Chemistry Question Paper Set 3 2025 with Answers

Inductive Effect and Its Role in Acidic Strength

Degree of Dissociation: Meaning, Formula, Calculation & Uses




