
The ABAB packing and ABCABC packing are respectively called as:
A. Hexagonal close packing (hcp) and cubic close packing (CCP)
B. Cubic close packing (ccp) and hexagonal close packing (HCP)
C. Body centred cubic (bcc) and hexagonal close packing (HCP)
D. Hexagonal close packing (hcp) and body centred cubic (BCC)
Answer
233.1k+ views
Hint: The different packing structures or types can be identified on the basis of the molecular arrangement in the given lattice. Each type of packing structure has a unique arrangement of atoms within them that can be understood by viewing the lattice from a side profile.
Complete Step-by-Step answer:
The spatial arrangements of atoms within a given lattice can be done in one dimension or two dimensions or even in 3 dimensions. Cubic close packing (ccp), hexagonal close packing (hcp) and body centred cubic (bcc) are all 3-dimensional spatial arrangements.
The spatial arrangements for the given packing structures are given as follows:
1. Hexagonal close packing
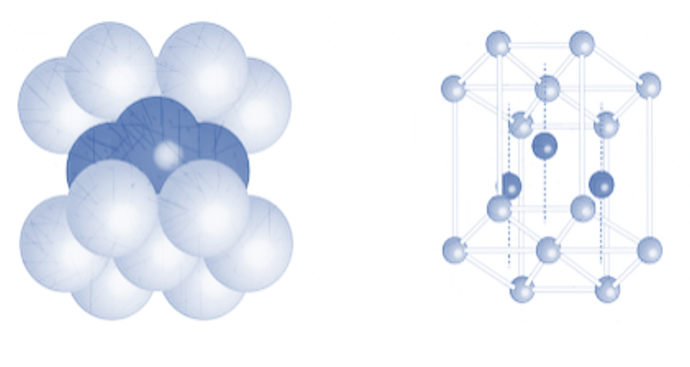
The arrangement in hexagonal packing as observed above is ABABAB
2. Cubic close packing
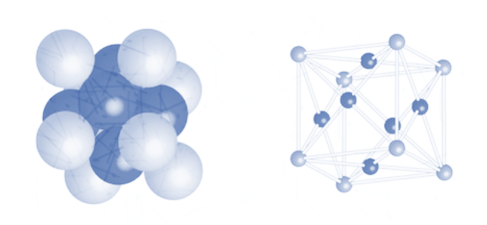
Face centred cubic packing is also known as cubic close packing. The arrangement in hexagonal packing as observed above is ABCABC
3. Body centred cubic packing
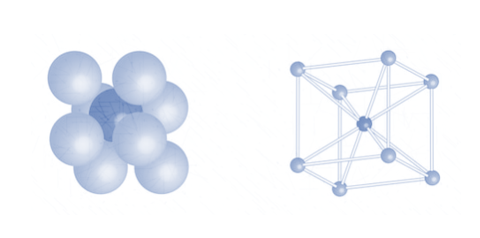
Since the body centric atoms are not in contact with any other atom within the same 2 – dimensional layer, it cannot be defined using a layer by layer interpretation.
Hence, the ABAB packing and ABCABC packing are respectively called as hexagonal close packing (hcp) and cubic close packing (ccp)
Hence, Option A is the correct option.
Note: The repetitive patterns of the different 2 - dimensional layers of the packing structures are named A, B, C and so on. So, each type of dimensional packing structure, has its own repetitive arrangements of the 2 - dimensional layers which can be represented in the above fashion.
Complete Step-by-Step answer:
The spatial arrangements of atoms within a given lattice can be done in one dimension or two dimensions or even in 3 dimensions. Cubic close packing (ccp), hexagonal close packing (hcp) and body centred cubic (bcc) are all 3-dimensional spatial arrangements.
The spatial arrangements for the given packing structures are given as follows:
1. Hexagonal close packing

The arrangement in hexagonal packing as observed above is ABABAB
2. Cubic close packing

Face centred cubic packing is also known as cubic close packing. The arrangement in hexagonal packing as observed above is ABCABC
3. Body centred cubic packing

Since the body centric atoms are not in contact with any other atom within the same 2 – dimensional layer, it cannot be defined using a layer by layer interpretation.
Hence, the ABAB packing and ABCABC packing are respectively called as hexagonal close packing (hcp) and cubic close packing (ccp)
Hence, Option A is the correct option.
Note: The repetitive patterns of the different 2 - dimensional layers of the packing structures are named A, B, C and so on. So, each type of dimensional packing structure, has its own repetitive arrangements of the 2 - dimensional layers which can be represented in the above fashion.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions (2025-26)

Solutions Class 12 Chemistry Chapter 1 CBSE Notes - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The d and f Block Elements (2025-26)

Biomolecules Class 12 Chemistry Chapter 10 CBSE Notes - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules (2025-26)




