
Cyclohexanone on being treated with NaOH solution forms:
A.
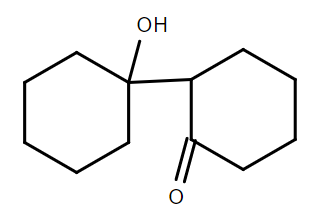
B.
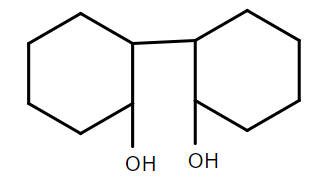
C.
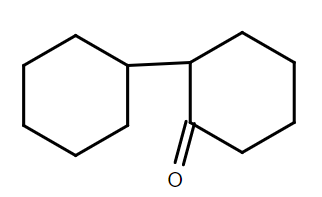
D.
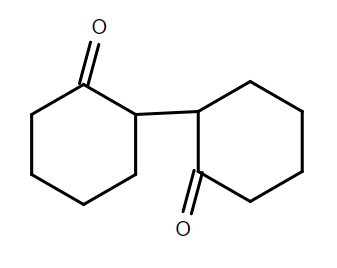




Answer
576.3k+ views
Hint:To answer this question, you should recall the concept of an aldol condensation. The reaction of aldehydes and ketones containing at least one hydrogen is treated with dilute alkali; they form -hydroxy aldehydes or -hydroxy ketones (ketol) respectively.
Complete step by step answer:
We know that in aldol condensation the hydroxide ion functions as a base moving the acidic hydrogen-producing the reactive enolate ion. Further, the aldehyde is attacked at the electrophilic carbonyl carbon by the nucleophilic enolate ion resulting in an alkoxide intermediate. The alkoxide ion now formed deprotonates the water molecule, ultimately resulting in hydroxide and the β–hydroxy aldehyde.
The product obtained when cyclohexanone is heated with a NaOH solution is as shown by the following mechanism:
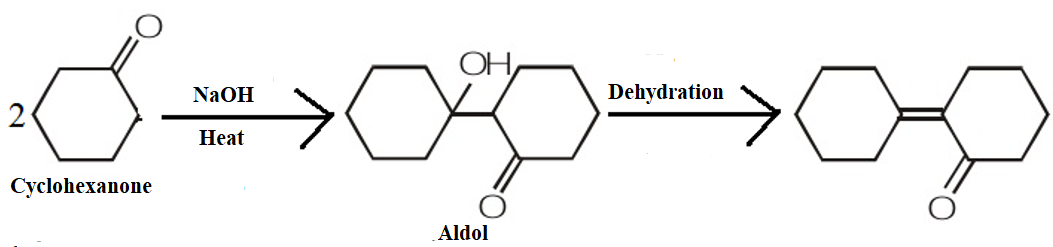
Therefore, we can conclude that the correct answer to this question is option C.
Note:
Make sure you remember the difference between aldol condensation and redox process. Aldox process can be termed as another variation of aldol condensation used in industries for the direct conversion of syngas and propene into 2-ethyl hexanol. The product's formation takes place with hydroformylation of the reactants into butyraldehyde, which further undergoes aldol condensation into 2-ethyl hexenal and the hydrogenation of this intermediate into 2-ethyl hexanol.
Complete step by step answer:
We know that in aldol condensation the hydroxide ion functions as a base moving the acidic hydrogen-producing the reactive enolate ion. Further, the aldehyde is attacked at the electrophilic carbonyl carbon by the nucleophilic enolate ion resulting in an alkoxide intermediate. The alkoxide ion now formed deprotonates the water molecule, ultimately resulting in hydroxide and the β–hydroxy aldehyde.
The product obtained when cyclohexanone is heated with a NaOH solution is as shown by the following mechanism:

Therefore, we can conclude that the correct answer to this question is option C.
Note:
Make sure you remember the difference between aldol condensation and redox process. Aldox process can be termed as another variation of aldol condensation used in industries for the direct conversion of syngas and propene into 2-ethyl hexanol. The product's formation takes place with hydroformylation of the reactants into butyraldehyde, which further undergoes aldol condensation into 2-ethyl hexenal and the hydrogenation of this intermediate into 2-ethyl hexanol.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




