
Draw the orbit structure to show the formation of the following:
A.Oxygen molecule
B.Ammonia
C.Calcium Oxide
(\[\text{H = 1}\], $\text{N = 7}$, $\text{O = 8}$, $\text{Ca = 20}$)
Answer
560.4k+ views
Hint: The arrangement of all the electrons in an atom of an element or the molecule of a compound in all its orbits is known as the orbit structure of the element or the compound.
Complete answer:
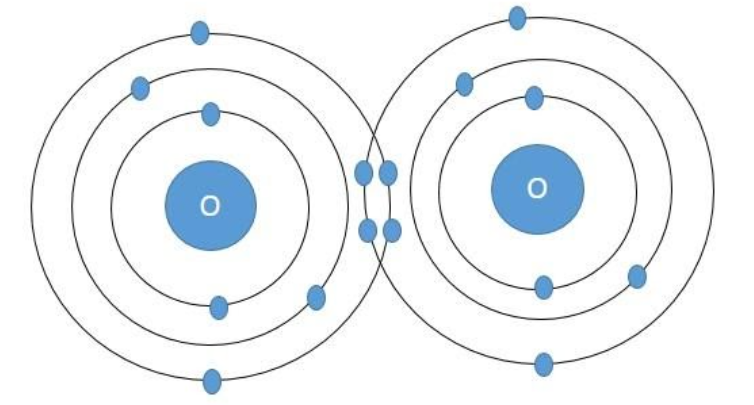
Oxygen molecule – In the atom of oxygen, there are total eight electrons arranged in the electronic configuration:$\text{1}{{\text{s}}^{\text{2}}}\text{2}{{\text{s}}^{\text{2}}}\text{2}{{\text{p}}^{\text{4}}}$. Accordingly, there are a total of six electrons in the valence shell of the oxygen atom and it is short of two electrons to attain the octet confirmation. So the oxygen atom shares two electrons with another oxygen atom to form the oxygen molecule according to the following diagram.

Ammonia – In the ammonia molecule, there is one nitrogen atom and three hydrogen atoms. The electronic configuration of nitrogen is $\text{1}{{\text{s}}^{\text{2}}}\text{2}{{\text{s}}^{\text{2}}}\text{2}{{\text{p}}^{3}}$and that for hydrogen is$\text{1}{{\text{s}}^{\text{1}}}$. Hence hydrogen is one electron short of duplet and nitrogen is three electrons short of an octet. So the nitrogen forms three covalent bonds with hydrogen atoms to form ammonia.
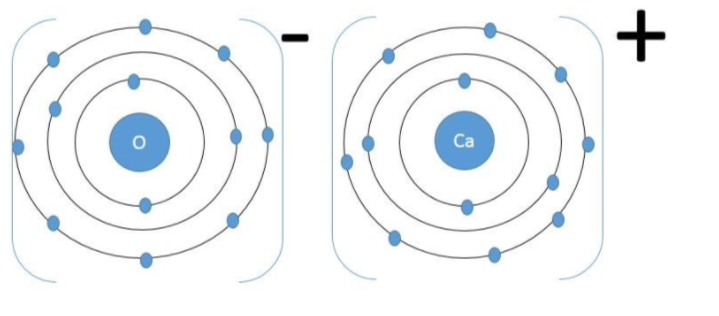
Calcium Oxide – The electronic configuration of calcium is$\text{1}{{\text{s}}^{\text{2}}}\text{2}{{\text{s}}^{\text{2}}}\text{2}{{\text{p}}^{6}}\text{3}{{\text{s}}^{\text{2}}}\text{3}{{\text{p}}^{\text{6}}}\text{4}{{\text{s}}^{\text{2}}}$. So the calcium atom donates two of its valence shell electrons to form the calcium dipositive cation while the oxygen atom takes up those electrons to form the oxide anion and together they form the calcium oxide ionic solid.
Note:
The atoms gain or lose electrons to attain a stable electronic confirmation. But when the number of electrons required to attain the octet configuration is too large, then the atoms prefer to share the electrons and form covalent bonds instead of gaining or losing electrons.
Complete answer:
Oxygen molecule – In the atom of oxygen, there are total eight electrons arranged in the electronic configuration:$\text{1}{{\text{s}}^{\text{2}}}\text{2}{{\text{s}}^{\text{2}}}\text{2}{{\text{p}}^{\text{4}}}$. Accordingly, there are a total of six electrons in the valence shell of the oxygen atom and it is short of two electrons to attain the octet confirmation. So the oxygen atom shares two electrons with another oxygen atom to form the oxygen molecule according to the following diagram.

Ammonia – In the ammonia molecule, there is one nitrogen atom and three hydrogen atoms. The electronic configuration of nitrogen is $\text{1}{{\text{s}}^{\text{2}}}\text{2}{{\text{s}}^{\text{2}}}\text{2}{{\text{p}}^{3}}$and that for hydrogen is$\text{1}{{\text{s}}^{\text{1}}}$. Hence hydrogen is one electron short of duplet and nitrogen is three electrons short of an octet. So the nitrogen forms three covalent bonds with hydrogen atoms to form ammonia.

Calcium Oxide – The electronic configuration of calcium is$\text{1}{{\text{s}}^{\text{2}}}\text{2}{{\text{s}}^{\text{2}}}\text{2}{{\text{p}}^{6}}\text{3}{{\text{s}}^{\text{2}}}\text{3}{{\text{p}}^{\text{6}}}\text{4}{{\text{s}}^{\text{2}}}$. So the calcium atom donates two of its valence shell electrons to form the calcium dipositive cation while the oxygen atom takes up those electrons to form the oxide anion and together they form the calcium oxide ionic solid.

Note:
The atoms gain or lose electrons to attain a stable electronic confirmation. But when the number of electrons required to attain the octet configuration is too large, then the atoms prefer to share the electrons and form covalent bonds instead of gaining or losing electrons.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




