
What is the packing efficiency for an end centered unit cell?
Answer
522.1k+ views
Hint- The packing efficiency can be calculated by the percent of space occupied by spheres present in a unit cell. Here we will proceed further by evaluating the volume of spheres in the unit cell and total volume of the unit cell.
Complete answer:
We will use the following figure of the end centered unit cell to solve the problem.
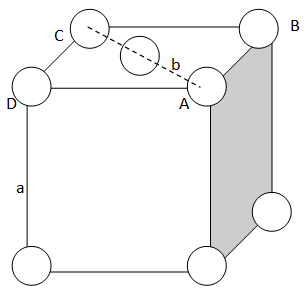
Let the side of an unit cell = a
And diagonal AC = b
Now, in right triangle ABC,
Let us use the Pythagoras theorem.
AD is perpendicular, DC is base and AC is diagonal
$
\because A{C^2} = A{D^2} + D{C^2} \\
\Rightarrow {b^2} = {a^2} + {a^2} \\
\Rightarrow {b^2} = 2{a^2} \\
\Rightarrow b = \sqrt 2 a \\
$
Let r is the radius of sphere, so b = 4r,
Thus,
\[
b = 4r = a\sqrt 2 \\
\Rightarrow a = \dfrac{{4r}}{{\sqrt 2 }} \\
\Rightarrow a = \dfrac{{4r}}{{\sqrt 2 }} \times \dfrac{{\sqrt 2 }}{{\sqrt 2 }} \\
\Rightarrow a = \dfrac{{4\sqrt 2 r}}{2} \\
\Rightarrow a = 2\sqrt 2 r.........(1) \\
\]
Now, volume of cube $ = {\left( {{\text{side}}} \right)^3} = {a^3}$
Substituting the value of a from equation (i) we get,
Volume of cube
$
= {a^3} = {\left( {2\sqrt 2 r} \right)^3} \\
= 8 \times 2\sqrt 2 \times {r^3} \\
= 16\sqrt 2 {r^3} \\
$
Volume of cube $ = 16\sqrt 2 {r^3}$ ---- (2)
Now, volume of sphere
$ = \dfrac{4}{3}\pi {r^3}..........(3)$
Since one unit cell of end entered cell has 2 spheres
$ = \dfrac{1}{8} \times 8 + \dfrac{1}{2} \times 2 = 1 + 1 = 2$
As the contribution of the corner sphere in a cell is one eighth and that of the face sphere is half.
Therefore, volume of 2 atoms, i.e. 2 spheres:
$
= 2 \times \dfrac{4}{3}\pi {r^3} \\
= \dfrac{8}{3}\pi {r^3} \\
$
We know that
Packing efficiency = (Volume of spheres in unit cell/ total volume of unit cell) × 100%
Since there are 2 atoms in the unit cell of end centered cell
Therefore, packing efficiency of end centered cell
Packing efficiency = (Volume of 2 spheres in unit cell/ total volume of unit cell) × 100%
Now, packing efficiency (in %)
$
= \dfrac{{{\text{volume of 2 spheres in unit cell}}}}{{{\text{total volume of unit cell}}}} \times 100 \\
= \dfrac{{\dfrac{8}{3}\pi {r^3}}}{{16\sqrt 2 {r^3}}} \times 100 \\
$
Let us solve the equation by cancelling the common term to find the percentage.
$
= \dfrac{{8\pi }}{{3 \times 16\sqrt 2 }} \times 100 \\
= \dfrac{{\pi \times 100}}{{3 \times 2\sqrt 2 }} \\
= \dfrac{{3.14 \times 50}}{{3 \times 1.414}} \\
= 37.02\% \\
$
Hence, packing efficiency for end centered unit cell is 37.02%
Note- Packing efficiency is defined as the percentage of space occupied by constituent particles packed inside the lattice. It can be calculated with the help of geometry in three structures namely: HCP and CCP structures. The packing efficiency of simple cubic lattice is 52.4%. And the packing efficiency of body centered cubic lattice (bcc) is 68%.
Complete answer:
We will use the following figure of the end centered unit cell to solve the problem.

Let the side of an unit cell = a
And diagonal AC = b
Now, in right triangle ABC,
Let us use the Pythagoras theorem.
AD is perpendicular, DC is base and AC is diagonal
$
\because A{C^2} = A{D^2} + D{C^2} \\
\Rightarrow {b^2} = {a^2} + {a^2} \\
\Rightarrow {b^2} = 2{a^2} \\
\Rightarrow b = \sqrt 2 a \\
$
Let r is the radius of sphere, so b = 4r,
Thus,
\[
b = 4r = a\sqrt 2 \\
\Rightarrow a = \dfrac{{4r}}{{\sqrt 2 }} \\
\Rightarrow a = \dfrac{{4r}}{{\sqrt 2 }} \times \dfrac{{\sqrt 2 }}{{\sqrt 2 }} \\
\Rightarrow a = \dfrac{{4\sqrt 2 r}}{2} \\
\Rightarrow a = 2\sqrt 2 r.........(1) \\
\]
Now, volume of cube $ = {\left( {{\text{side}}} \right)^3} = {a^3}$
Substituting the value of a from equation (i) we get,
Volume of cube
$
= {a^3} = {\left( {2\sqrt 2 r} \right)^3} \\
= 8 \times 2\sqrt 2 \times {r^3} \\
= 16\sqrt 2 {r^3} \\
$
Volume of cube $ = 16\sqrt 2 {r^3}$ ---- (2)
Now, volume of sphere
$ = \dfrac{4}{3}\pi {r^3}..........(3)$
Since one unit cell of end entered cell has 2 spheres
$ = \dfrac{1}{8} \times 8 + \dfrac{1}{2} \times 2 = 1 + 1 = 2$
As the contribution of the corner sphere in a cell is one eighth and that of the face sphere is half.
Therefore, volume of 2 atoms, i.e. 2 spheres:
$
= 2 \times \dfrac{4}{3}\pi {r^3} \\
= \dfrac{8}{3}\pi {r^3} \\
$
We know that
Packing efficiency = (Volume of spheres in unit cell/ total volume of unit cell) × 100%
Since there are 2 atoms in the unit cell of end centered cell
Therefore, packing efficiency of end centered cell
Packing efficiency = (Volume of 2 spheres in unit cell/ total volume of unit cell) × 100%
Now, packing efficiency (in %)
$
= \dfrac{{{\text{volume of 2 spheres in unit cell}}}}{{{\text{total volume of unit cell}}}} \times 100 \\
= \dfrac{{\dfrac{8}{3}\pi {r^3}}}{{16\sqrt 2 {r^3}}} \times 100 \\
$
Let us solve the equation by cancelling the common term to find the percentage.
$
= \dfrac{{8\pi }}{{3 \times 16\sqrt 2 }} \times 100 \\
= \dfrac{{\pi \times 100}}{{3 \times 2\sqrt 2 }} \\
= \dfrac{{3.14 \times 50}}{{3 \times 1.414}} \\
= 37.02\% \\
$
Hence, packing efficiency for end centered unit cell is 37.02%
Note- Packing efficiency is defined as the percentage of space occupied by constituent particles packed inside the lattice. It can be calculated with the help of geometry in three structures namely: HCP and CCP structures. The packing efficiency of simple cubic lattice is 52.4%. And the packing efficiency of body centered cubic lattice (bcc) is 68%.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




